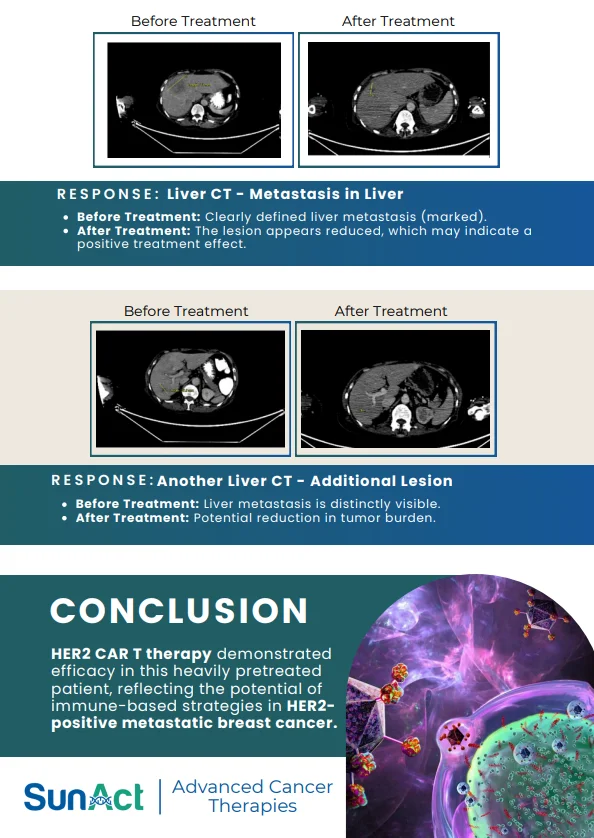
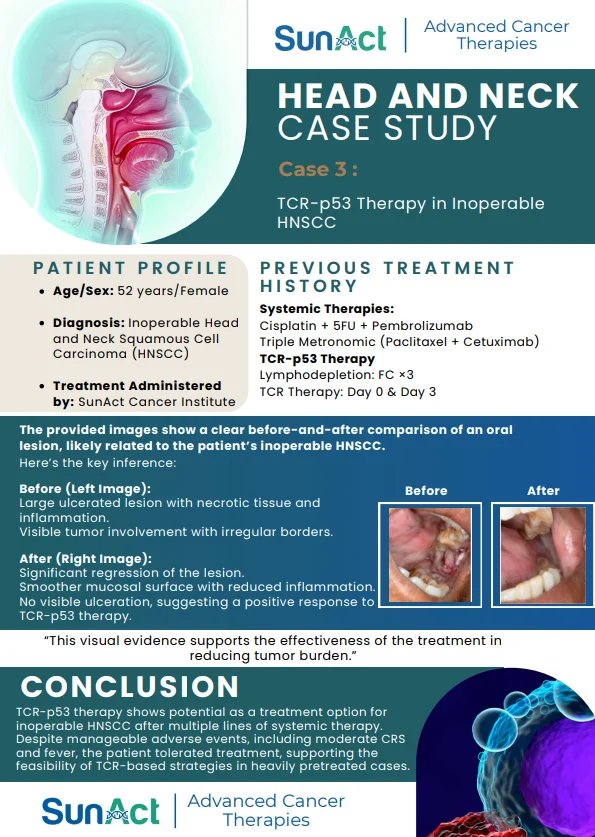
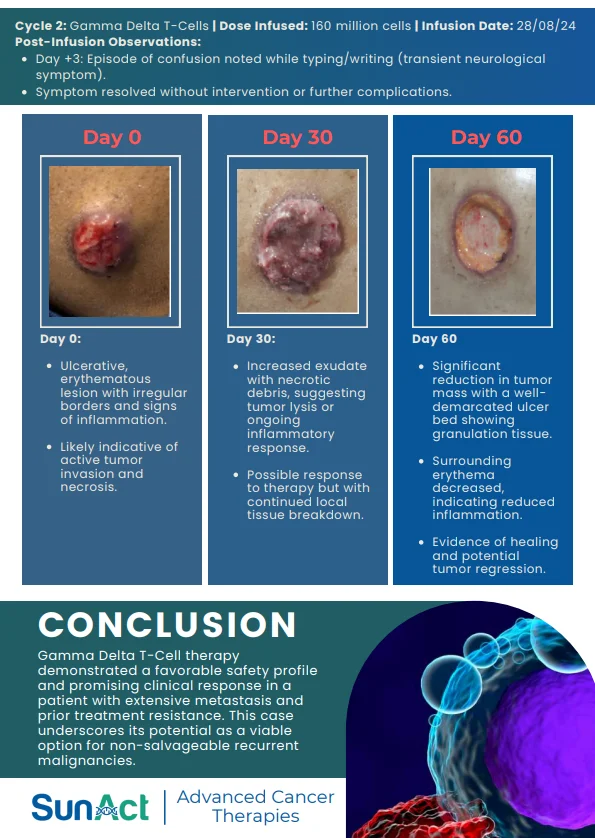
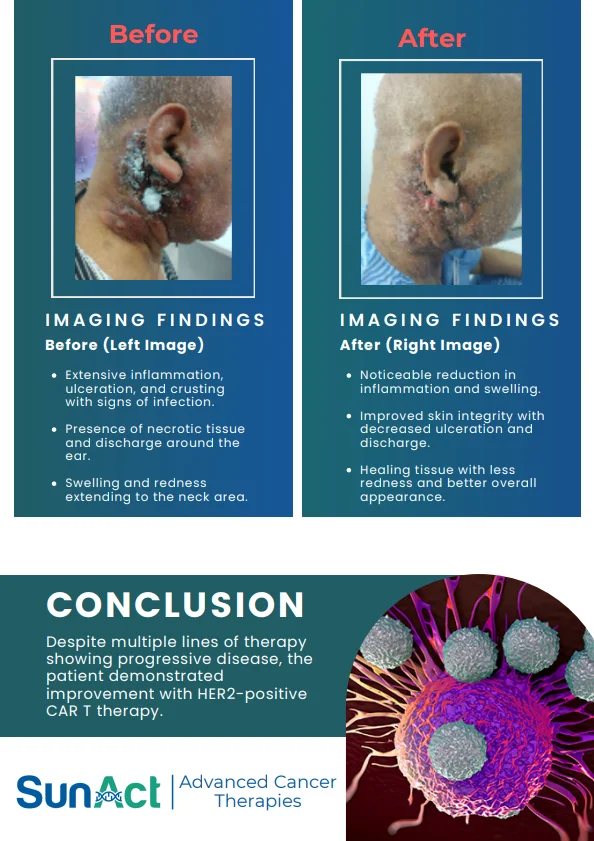
IN-VIVO CAR ENGINEERING | KRAS G12D DEGRADER TRIAL | SMM EARLY TREATMENT APPROVAL | AI NANO LIQUID BIOPSY
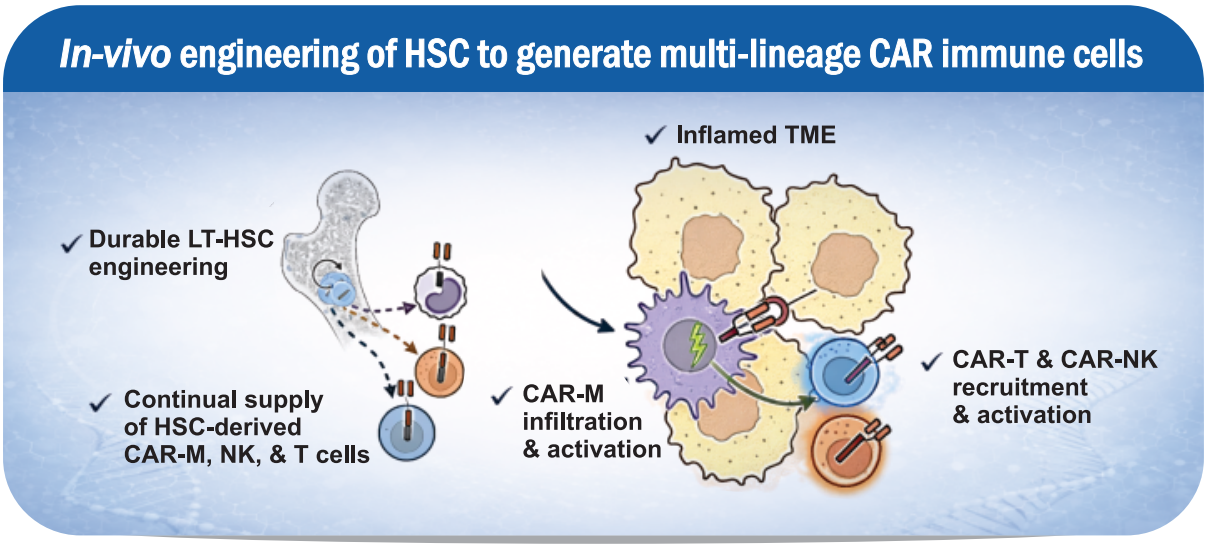
This study demonstrates a novel in-vivo molecular CAR therapy in which hematopoietic stem cells (HSCs) are genetically engineered directly in the body using a high-capacity virus-like particle (VLP) platform to generate lineage-restricted HER2 CAR macrophages, CAR-NK cells, and CAR-T cells. By targeting CD46-expressing primitive HSCs and enabling stable transgene integration, a single in-vivo intervention produced durable, multi-lineage CAR immune cells for at least five months, with evidence of long-term HSC engraftment shown by secondary transplantation. Functionally, these HSC-derived CAR-M, NK, and T cells cooperated to infiltrate tumors, remodel the tumor microenvironment, and suppress solid tumor growth in immunocompetent mouse models.
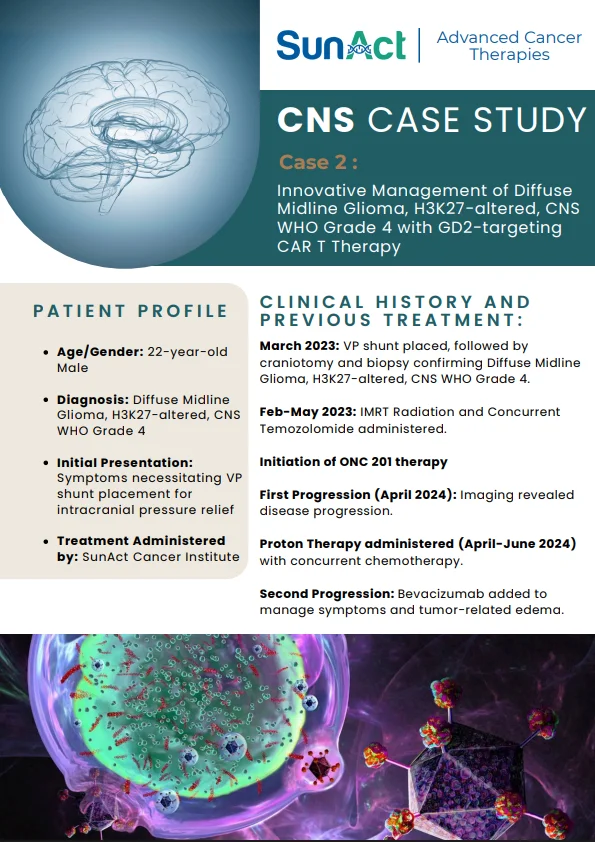
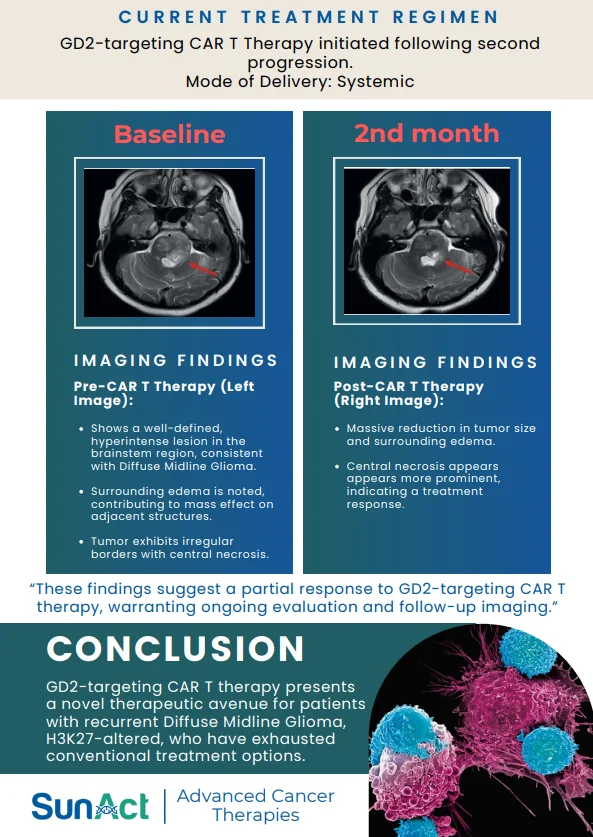
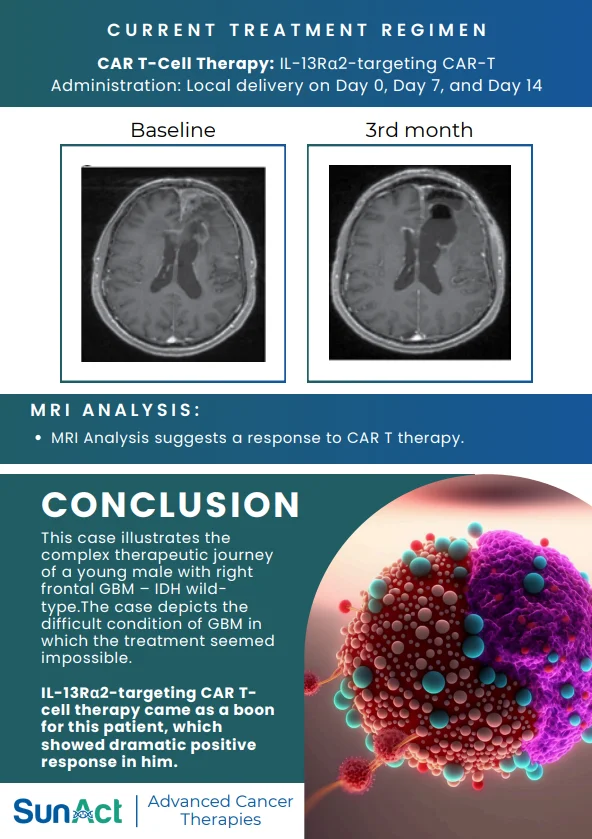
Reference: Zhou Y et al., J. Immunother Cancer 2023;11:e005142
ARV-806 is a first-in-class proteolysis targeting chimera (PROTAC) designed to selectively degrade KRAS G12D, the most prevalent KRAS mutation in cancer and a major unmet medical need in pancreatic, colorectal, and lung cancers. Unlike conventional KRAS inhibitors, ARV-806 induces proteasomal degradation of both GDP- and GTP-bound KRAS G12D, robustly reducing mutant KRAS levels and inhibiting downstream signaling. In preclinical models, ARV-806 demonstrated strong tumor regression in xenograft and PDX models supporting further clinical evaluation. Early results from a phase 1/2 trial (NCT07023731) are ongoing, evaluating pharmacokinetics, safety, and preliminary efficacy in patients with KRAS G12D-mutant advanced solid tumors, with a focus on pancreatic ductal adenocarcinoma (PDAC).
Reference: Mucins-Govil T et al., Clin Oncol 2024;42(3):TPS792
On January 27, 2026, daratumumab and hyaluronidase (Darzalex Faspro) became the first FDA-approved therapy for high-risk smouldering multiple myeloma (SMM). SMM has historically been managed with observation alone despite a substantial risk of progression to active multiple myeloma. This approval marks a major shift toward early therapeutic intervention. Daratumumab, an anti-CD38 monoclonal antibody, induces immune-mediated death of malignant plasma cells, while hyaluronidase enables subcutaneous delivery by enhancing tissue permeability, improving convenience and feasibility for long-term treatment in asymptomatic patients.

Reference: AACR, FDA Approvals in Oncology: October–December 2025
A group of scientists at the Zhejiang University, China has developed a novel technology for disease detection using liquid biopsy. This work introduces an intelligent nano-fingerprinting strategy for liquid biopsy that leverages single-molecule nano-pore technology combined with AI-based classification to analyse complex plasma matrices holistically. Instead of targeting predefined biomarkers, the approach captures global molecular fingerprints of blood in a label-free, amplification-free, and minimal-processing manner. Using only microlitre-scale volumes and simple dilution, nano-pore signals reflecting the intrinsic molecular heterogeneity of plasma are generated and accurately classified by intelligent algorithms, enabling efficient and potentially scalable disease detection.
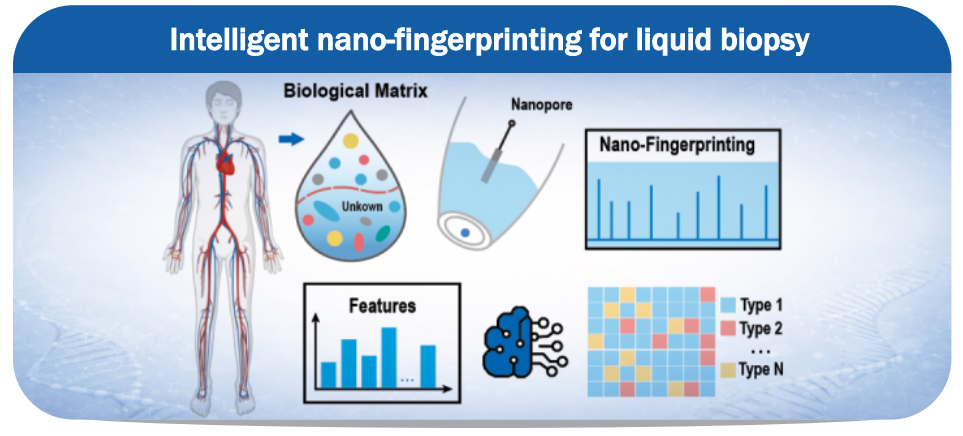
Reference: Yuxin Yang et. al., 2026, physics.bio-ph doi.org/10.48550/arXiv.2601.11947

At SunAct, we remain dedicated to tracking and sharing global advances that continue to redefine the landscape of cellular therapy and oncology. Stay tuned for our next edition as we uncover more breakthroughs and emerging trends shaping the future of cancer research.
Disclaimer: This newsletter is intended for healthcare professionals and researchers. Information is for educational purposes only.