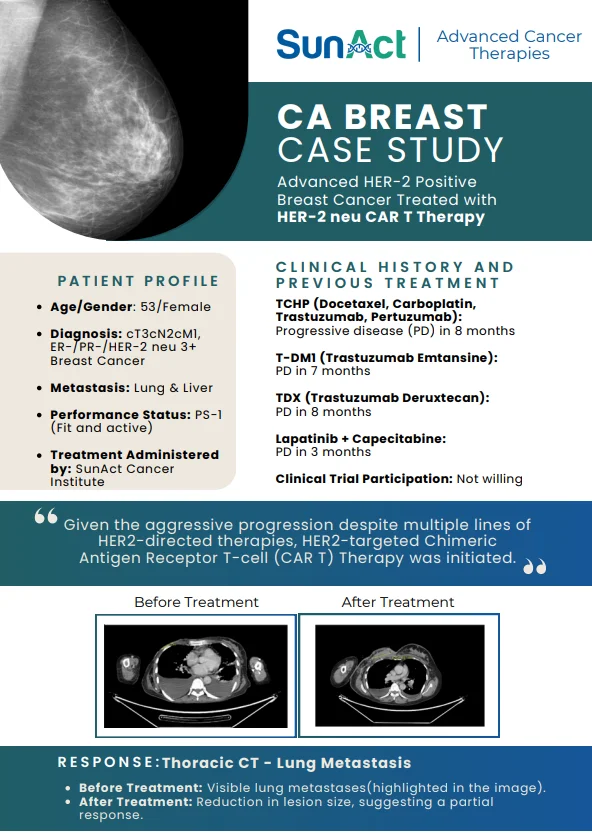
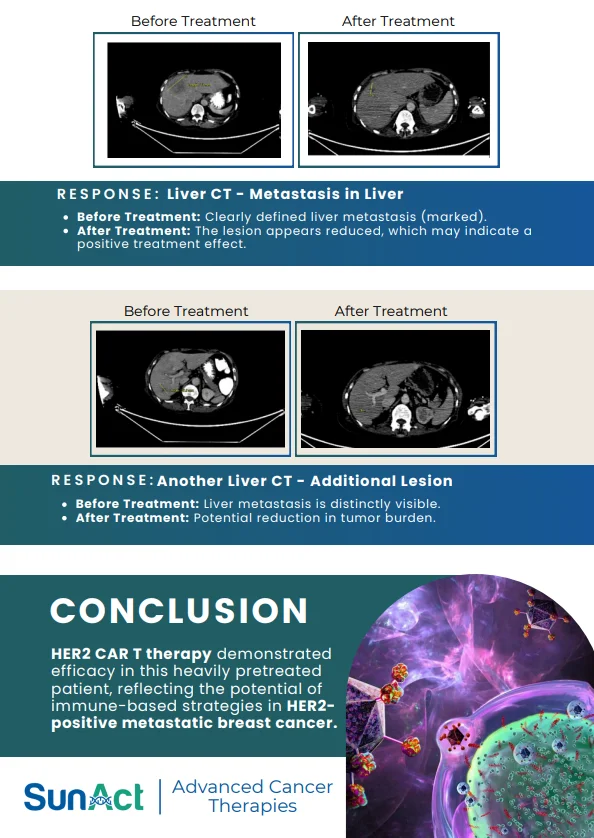
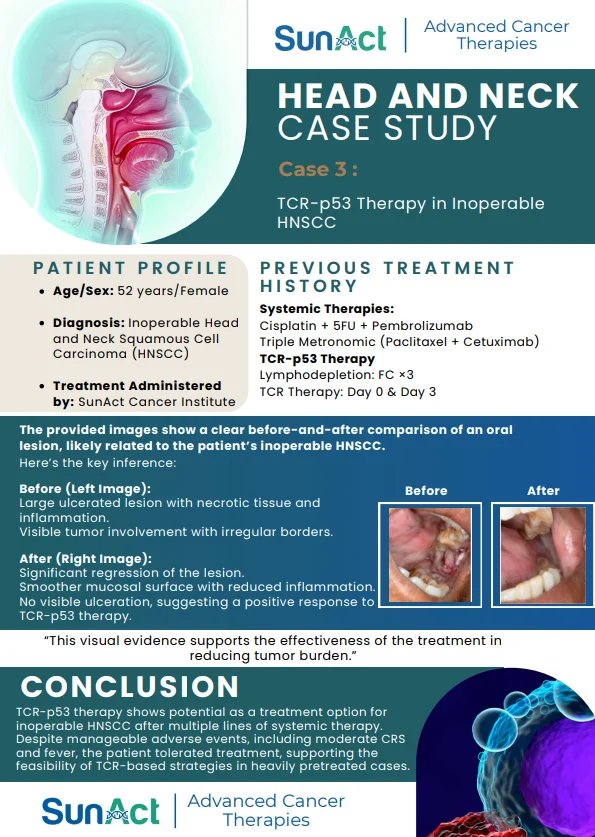
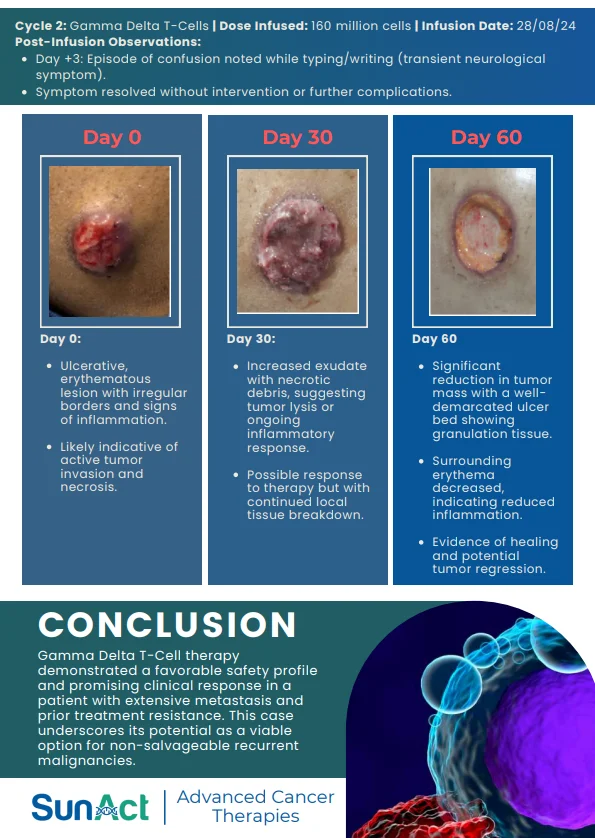
9 February 2026
Acute myeloid leukemia announces itself dramatically. Unlike chronic leukemias that develop slowly over months or years, this aggressive blood cancer progresses within weeks, demanding immediate attention and intensive treatment. Understanding its urgency while also recognizing the hope that modern treatments offer helps patients and families navigate this challenging diagnosis.
The disease begins in the bone marrow when young developing cells called myeloid blasts become cancerous. Instead of maturing into normal white blood cells, red blood cells, or platelets, these blasts multiply rapidly while remaining immature and nonfunctional. They crowd out healthy cells, leaving patients without adequate infection-fighting white cells, oxygen-carrying red cells, or clot-forming platelets.
Symptoms develop quickly and can be severe. Patients often present with profound fatigue and weakness from anemia. Easy bruising, bleeding gums, or nosebleeds signal low platelet counts. Fever and frequent infections indicate depleted normal white blood cells. Some patients notice bone pain or swollen gums. Others develop headaches, confusion, or breathlessness if leukemia cells infiltrate the brain or lungs.
Acute myeloid leukemia can affect anyone but becomes more common with advancing age. Most patients are older adults, though children and younger adults certainly develop the disease as well. Risk factors include previous chemotherapy or radiation, certain genetic syndromes, and pre-existing bone marrow disorders. However, most cases arise without an identifiable cause.
Diagnosis requires urgent blood tests revealing low normal blood cells and the presence of blast cells. Bone marrow examination confirms the diagnosis, with pathologists identifying blast cells exceeding 20 percent of bone marrow cells. Crucially, detailed genetic and molecular testing classifies acute myeloid leukemia into subtypes that dramatically influence prognosis and treatment approach.
These genetic categories matter enormously. Favorable-risk leukemias, with specific genetic changes such as core-binding factor abnormalities, respond well to chemotherapy and have cure rates approaching 60 to 70 percent. Intermediate-risk disease, which is the largest category, shows variable outcomes. Adverse-risk leukemia, characterized by complex genetic changes or specific high-risk mutations, proves more difficult to treat and often requires bone marrow transplant for cure.
Treatment begins immediately upon diagnosis. Induction chemotherapy is intensive treatment given in the hospital and aims to destroy leukemia cells and achieve remission. The standard regimen combines cytarabine and an anthracycline drug, administered over seven days. Patients remain hospitalized for three to four weeks while blood counts recover, requiring transfusions, antibiotics, and intensive supportive care.
About 60 to 75 percent of younger patients achieve complete remission after induction, meaning bone marrow examination shows no detectable leukemia. Older or frail patients have lower remission rates but still derive benefit from treatment.
Remission, however, is not cure. Without further treatment, leukemia almost always returns. Consolidation therapy consists of additional chemotherapy cycles that deepen remission and reduce relapse risk. Patients with favorable-risk disease may achieve cure with chemotherapy alone. Those with intermediate- or adverse-risk leukemia often need allogeneic bone marrow transplant to maximize cure chances.
Transplant timing is crucial. Performing transplant in first remission offers the best outcomes for high-risk patients, with cure rates of 40 to 50 percent or higher. Waiting until relapse significantly reduces success rates.
New treatments have expanded options. Targeted therapies such as FLT3 inhibitors benefit patients whose leukemia carries FLT3 mutations, present in about 30 percent of cases. IDH inhibitors help those with IDH mutations. These drugs, combined with chemotherapy, improve outcomes substantially.
Older or frail patients who are unable to tolerate intensive chemotherapy may receive less aggressive treatment with hypomethylating agents such as azacitidine or venetoclax-based combinations. While less likely to cure, these gentler regimens can control disease and maintain quality of life.
Prognosis varies widely based on age, genetics, and treatment response. Younger patients with favorable genetics have cure rates of 60 to 70 percent. Adverse genetics or older age reduces this to 20 to 30 percent. However, even patients with difficult disease sometimes achieve long-term survival, and ongoing research continues to improve outcomes.
Living through acute myeloid leukemia treatment demands resilience. Hospitalizations are long, side effects are significant, and the emotional toll is substantial. However, modern supportive care manages symptoms effectively, and for many patients, intensive treatment leads to cure and a return to normal life without leukemia.
Acute myeloid leukemia is serious, urgent, and challenging. Yet with immediate action, expert care, and appropriate treatment, many patients overcome this disease and reclaim their future.