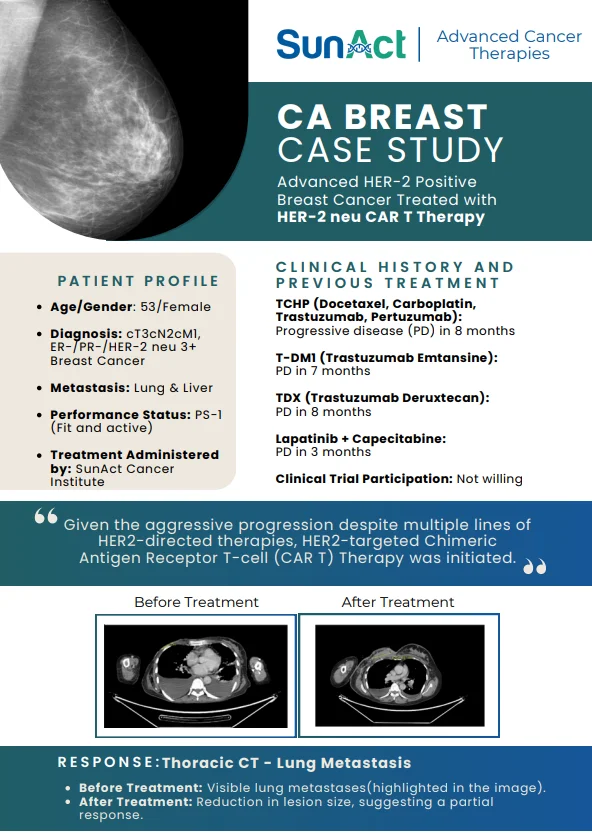
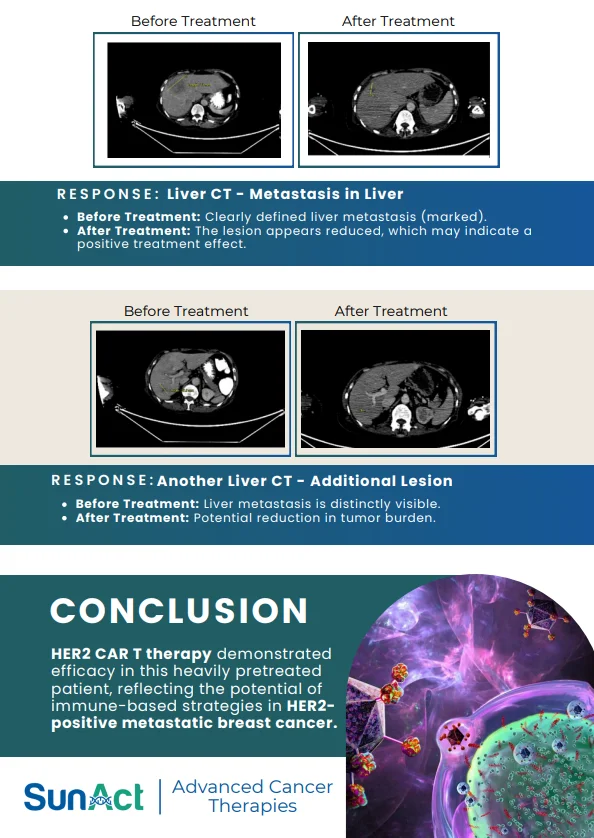
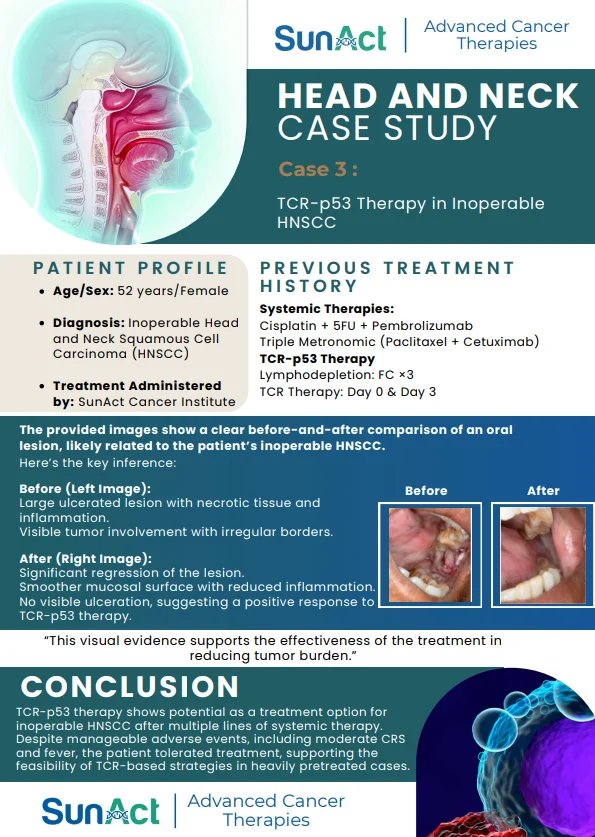
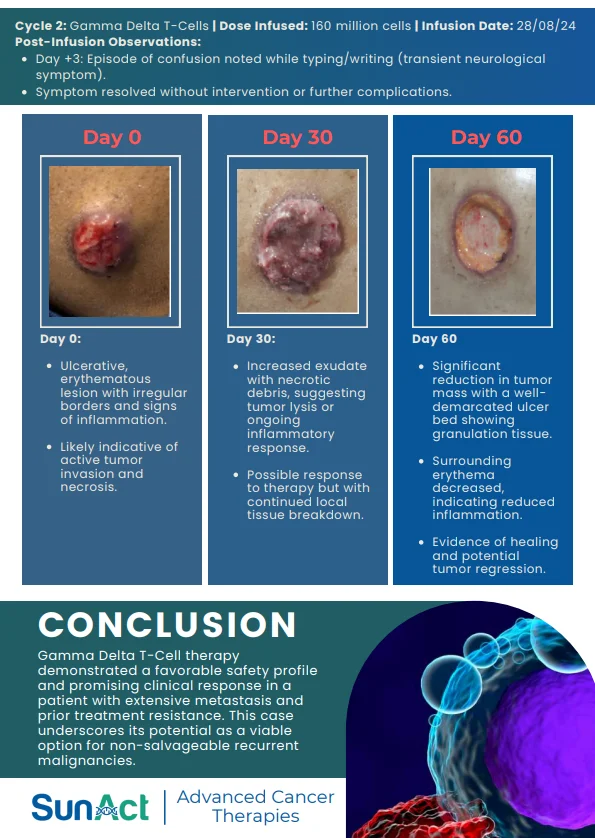
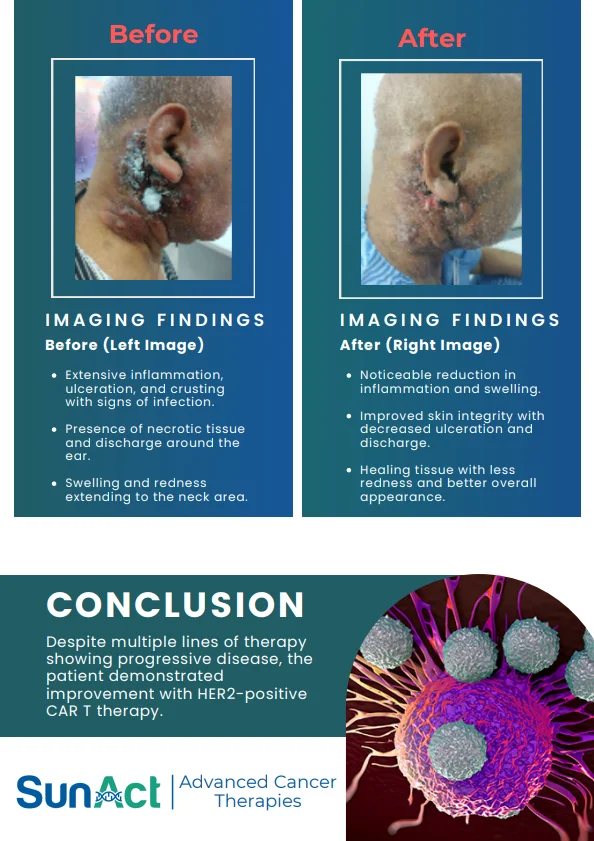
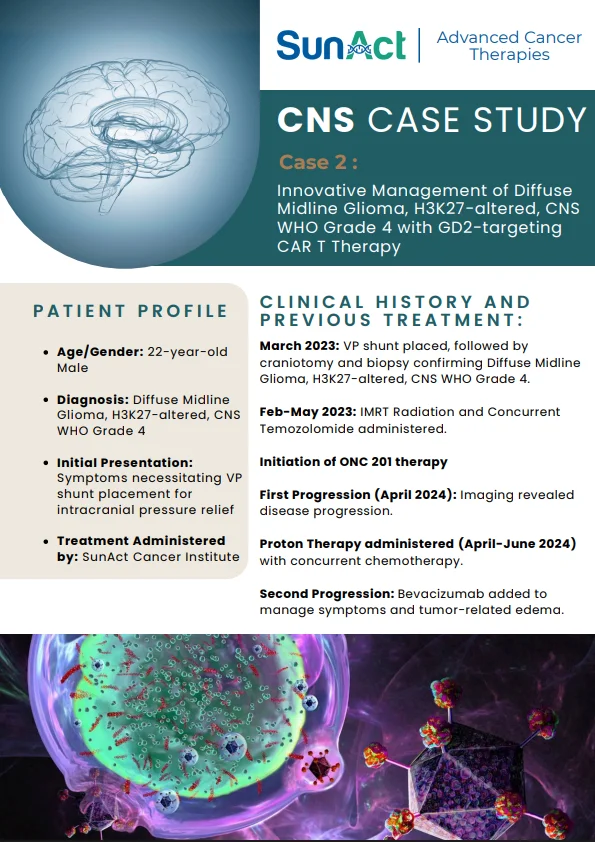
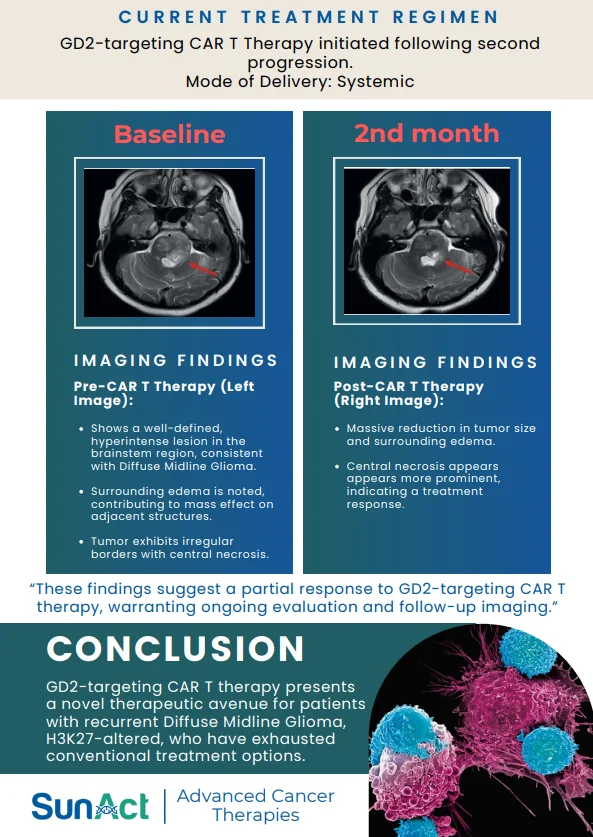
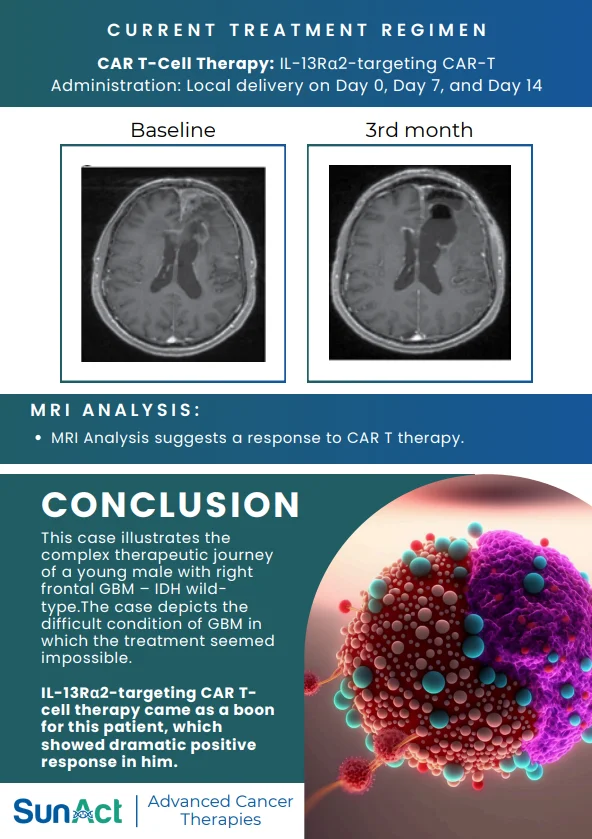
9 February 2026
Around twenty years ago, chronic myeloid leukemia was a devastating diagnosis. Patients faced a median survival of just three to five years, with bone marrow transplant offering the only chance for cure. Then came a revolution in targeted therapy, drugs that transformed this once-fatal cancer into a manageable chronic condition. Today, most patients with this disease live normal lifespans, taking a daily tablet that keeps their leukemia under control.

Chronic myeloid leukemia develops from a specific genetic accident. A piece of chromosome 9 swaps places with a piece of chromosome 22, creating an abnormal chromosome called the Philadelphia chromosome. This rearrangement produces a fusion gene that drives uncontrolled production of white blood cells, particularly granulocytes, the cells that fight bacterial infections.
The disease typically progresses through three phases. The chronic phase, where most patients are diagnosed, causes few symptoms initially. Blood tests reveal elevated white cell counts, often discovered incidentally during routine health checkups. Patients might notice fatigue, mild fever, night sweats, or a feeling of fullness due to an enlarged spleen. Some experience weight loss or feel generally unwell.
Without treatment, the disease eventually accelerates. The accelerated phase is marked by rising blast cells, which are immature white blood cells in the blood and bone marrow, worsening symptoms, and declining response to treatment. Finally, blast crisis resembles acute leukemia, with rapidly deteriorating health and poor outcomes. Modern treatment aims to prevent these later phases entirely.
Diagnosis requires blood tests showing characteristic patterns, including very high white cell counts with cells at various stages of maturation. Bone marrow examination confirms the diagnosis and establishes the disease phase. Crucially, testing for the Philadelphia chromosome or the BCR-ABL fusion gene is essential, as this genetic marker predicts response to targeted therapy.
Treatment has become remarkably straightforward for most patients. Tyrosine kinase inhibitors, drugs such as imatinib, dasatinib, or nilotinib, specifically block the abnormal protein produced by the BCR-ABL gene. Patients take one tablet daily, typically for life. These medications do not cure the disease in most cases, but they suppress it so effectively that leukemia cells become undetectable by standard tests.
The concept of treatment response has become central to management. Doctors monitor patients regularly with blood tests and sometimes bone marrow examinations. Complete hematological response means blood counts return to normal. Cytogenetic response indicates disappearance of Philadelphia chromosome positive cells. Molecular response, the deepest level, means BCR-ABL levels drop to very low or undetectable amounts.
Most patients achieve excellent responses. Studies show that over 80 percent of patients treated with modern tyrosine kinase inhibitors achieve deep molecular responses and enjoy survival rates approaching those of the general population. Some patients who maintain deep responses for several years may even attempt treatment-free remission by stopping medication under close supervision.
Side effects exist but are generally manageable. Imatinib commonly causes fluid retention, muscle cramps, nausea, or skin rashes. Dasatinib may cause fluid around the lungs. Nilotinib can affect glucose and lipid metabolism. However, most patients tolerate their medication well, adjusting doses or switching drugs if problems arise.
Regular monitoring ensures treatment works effectively. Initially, patients need monthly blood tests and periodic bone marrow examinations. Once stable, testing becomes less frequent, typically every three months for blood tests that check BCR-ABL levels. Any sign of rising leukemia markers prompts investigation for resistance and possible treatment adjustment.
Resistance remains a concern for some patients. Mutations in the BCR-ABL gene can make leukemia cells resistant to certain tyrosine kinase inhibitors. Fortunately, multiple drugs are available, and newer agents such as ponatinib overcome most resistance mutations. For the rare patient who fails multiple medications, bone marrow transplant may still offer curative potential.
Living with chronic myeloid leukemia has become remarkably normal. Patients work, travel, raise families, and pursue their goals while managing their condition with daily medication. The disease requires ongoing attention and monitoring, but for most, it no longer defines or limits their lives.
This transformation from a fatal disease to a chronic condition represents one of modern medicine’s greatest triumphs. It demonstrates that understanding cancer biology at the molecular level can lead to treatments that fundamentally change outcomes.