Novel Genetic Target for SCD | Low-Cost Detection of SCD and β-Thalassemia | New FDA Approval

SunAct is pleased to welcome Dr. Mamta Manglani as Director, Pediatric Hematology – Oncology & Bone Marrow Transplant. Dr. Manglani brings extensive expertise in pediatric cancers and hematological disorders, with special focus on hemoglobinopathies and a keen interest in pediatric bone marrow transplantation.
A landmark 2025 genome-wide association study (GWAS) published in Nature Communications has identified 14 novel candidate loci—most notably FLT1—that regulate fetal hemoglobin (HbF) levels in patients of African ancestry with Sickle Cell Disease (SCD). By analyzing a combined cohort of 3,751 individuals from Cameroon, Tanzania, and the USA, researchers demonstrated that these markers explain a staggering 94% of HbF heritability in African populations, a massive leap from the previously understood 10–24%.
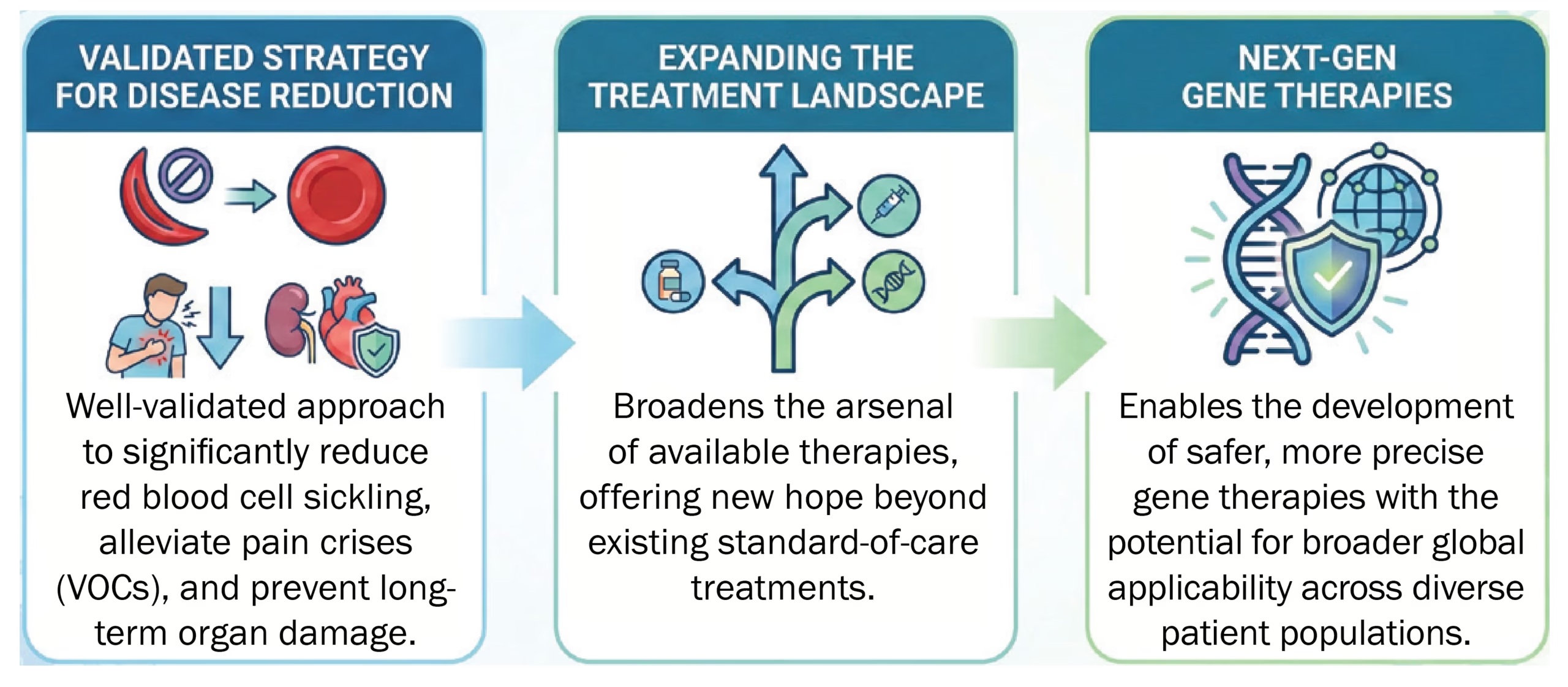
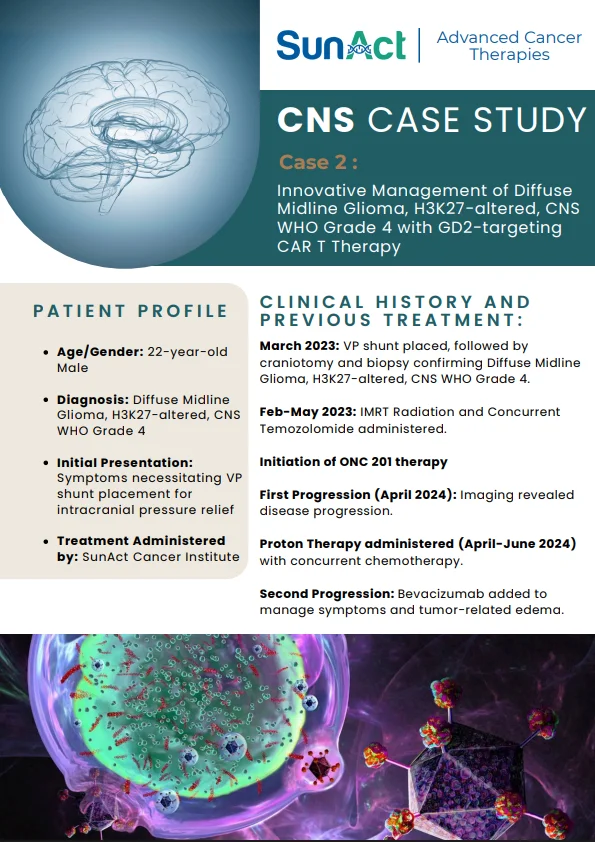
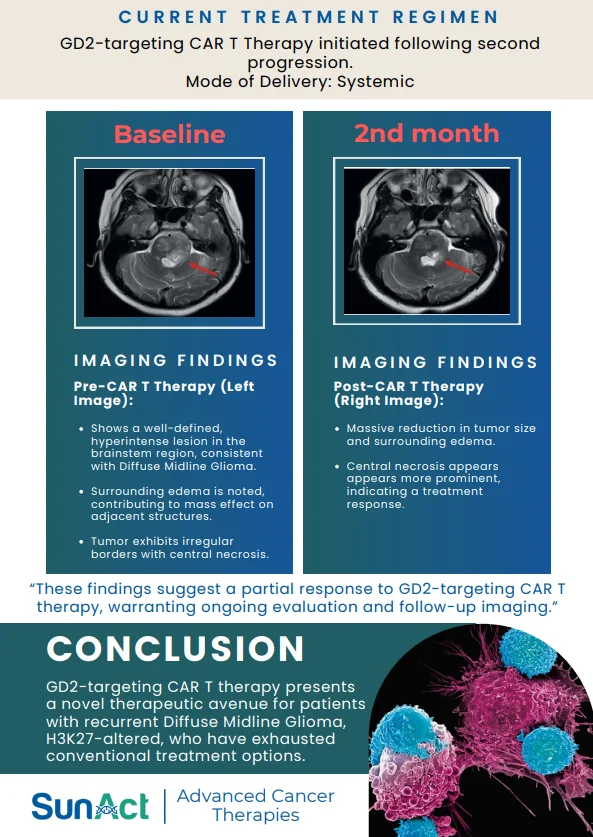
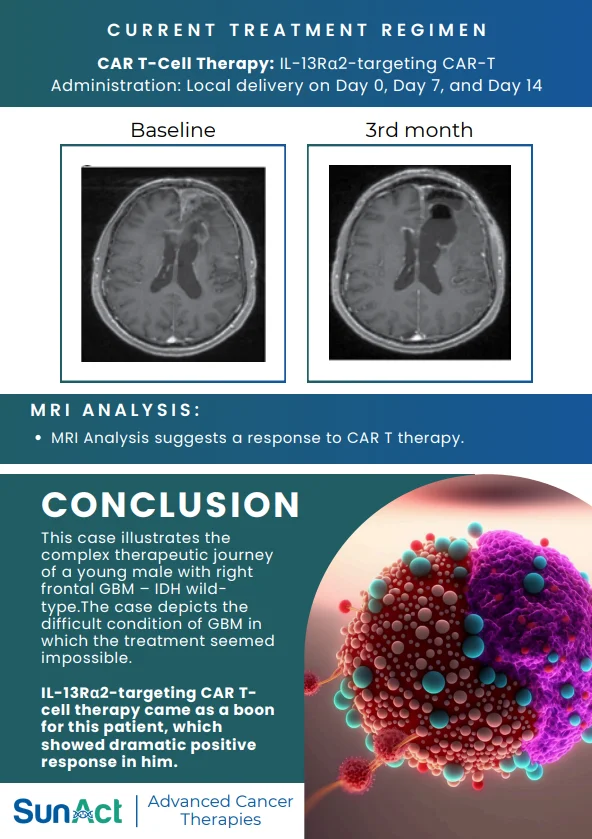
This discovery adds to the growing pipeline of genetic strategies aimed at providing durable and curative treatments for SCD, which affects approximately 300,000 newborns worldwide each year.
Source: Wonkam, A., Esoh, K., Levine, R.M. et al. FLT1 and other candidate fetal haemoglobin modifying loci in sickle cell disease in African ancestries. Nat Commun 2025;16:2092.
A landmark 2025 study published in The Lancet Regional Health – Southeast Asia evaluated affordable point-of-care devices for diagnosing sickle cell
disease (SCD) and β- thalassemia trait across 12 international sites in Africa,
Asia, and the Middle East. In this open-label, multicentre trial involving over 4,500 participants, tools like the Gazelle Hb Variant Test, HemoTypeSC, and automated sickling tests were evaluated against gold-standard HPLC methods. These innovations detect key hemoglobin variants (HbS, HbF, HbA) rapidly without needing labs, delivering results in under 10 minutes.
Devices achieved impressive metrics: Gazelle and HemoTypeSC offered 91–95% sensitivity and 98% specificity for SCD and carrier states, while sickling tests hit 89% accuracy overall.
A landmark 2025 study published in The Lancet Regional Health – Southeast Asia evaluated affordable point-of-care devices for diagnosing sickle cell disease (SCD) and β- thalassemia trait across 12 international sites in Africa,
Asia, and the Middle East. In this open-label, multicentre trial involving over 4,500 participants, tools like the Gazelle Hb Variant Test, HemoTypeSC, and automated sickling tests were evaluated against gold-standard HPLC methods. These innovations detect key hemoglobin variants (HbS, HbF, HbA) rapidly without needing labs, delivering results in under 10 minutes.
Devices achieved impressive metrics: Gazelle and HemoTypeSC offered 91–95% sensitivity and 98% specificity for SCD and carrier states, while sickling tests hit 89% accuracy overall. They performed reliably across diverse populations, though elevated fetal hemoglobin (HbF) occasionally caused interference. The trial highlights their potential for newborn and community screening in high-burden, resourcelimited areas.
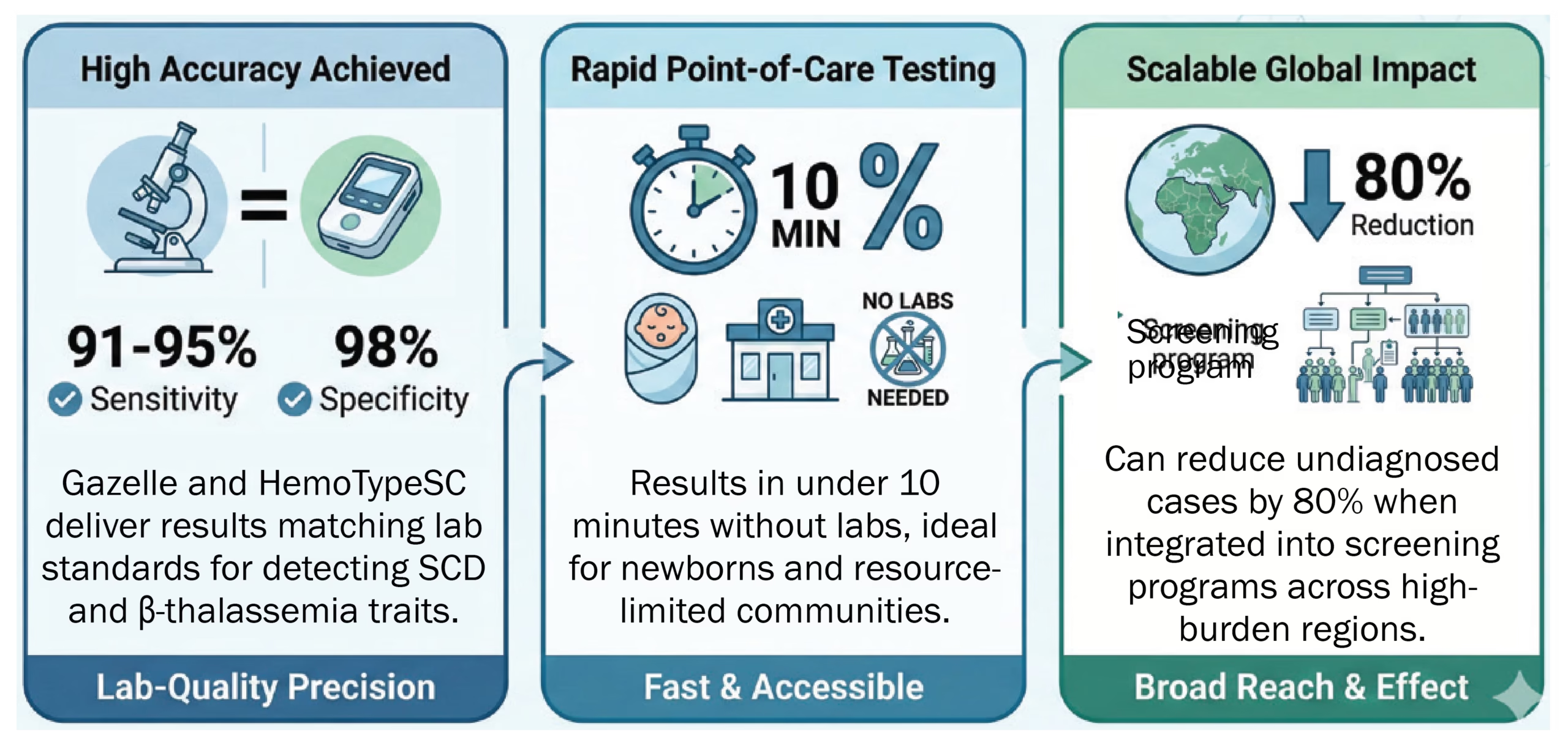
They performed reliably across diverse populations, though elevated fetal hemoglobin (HbF) occasionally caused interference. The trial highlights their potential for newborn and community screening in high-burden, resourcelimited areas.
Source: Shrestha P, Lohse H, Bhatla C, et al. Evaluation of low-cost techniques to detect sickle cell disease and β-thalassemia: an open-label, international, multicentre study. Lancet Reg
Health Southeast Asia. 2025;35:100571.
Fitusiran (Qfitlia) is a novel siRNA-based, non–factor rebalancing therapy approved by the USFDA in March 2025 for routine prophylaxis of
bleeding in patients aged ≥12 years with hemophilia A or B, with or
without inhibitors. Unlike factor replacement therapies, Fitusiran targets
hepatic antithrombin (AT) mRNA, leading to reduced antithrombin levels
and enhanced thrombin generation, thereby restoring hemostatic balance.
It is administered as a subcutaneous injection once every 4–8 months,
with individualized dose adjustment guided by antithrombin activity. Data
from the ATLAS phase 3 clinical program demonstrated marked reductions in annualized bleeding rates across both inhibitor and non-inhibitor
populations, highlighting its broad applicability. Safety monitoring is critical, as the therapy carries a boxed warning for thrombotic events and
Fitusiran (Qfitlia) is a novel siRNA-based, non–factor rebalancing therapy approved by the USFDA in March 2025 for routine prophylaxis of
bleeding in patients aged ≥12 years with hemophilia A or B, with or without inhibitors. Unlike factor replacement therapies, Fitusiran targets hepatic antithrombin (AT) mRNA, leading to reduced antithrombin levels and enhanced thrombin generation, thereby restoring hemostatic balance.
It is administered as a subcutaneous injection once every 4–8 months,
with individualized dose adjustment guided by antithrombin activity. Data
from the ATLAS phase 3 clinical program demonstrated marked reductions in annualized bleeding rates across both inhibitor and non-inhibitor populations, highlighting its broad applicability. Safety monitoring is critical, as the therapy carries a boxed warning for thrombotic events and gallbladder disease, along with the need for regular liver function and antithrombin level assessments.
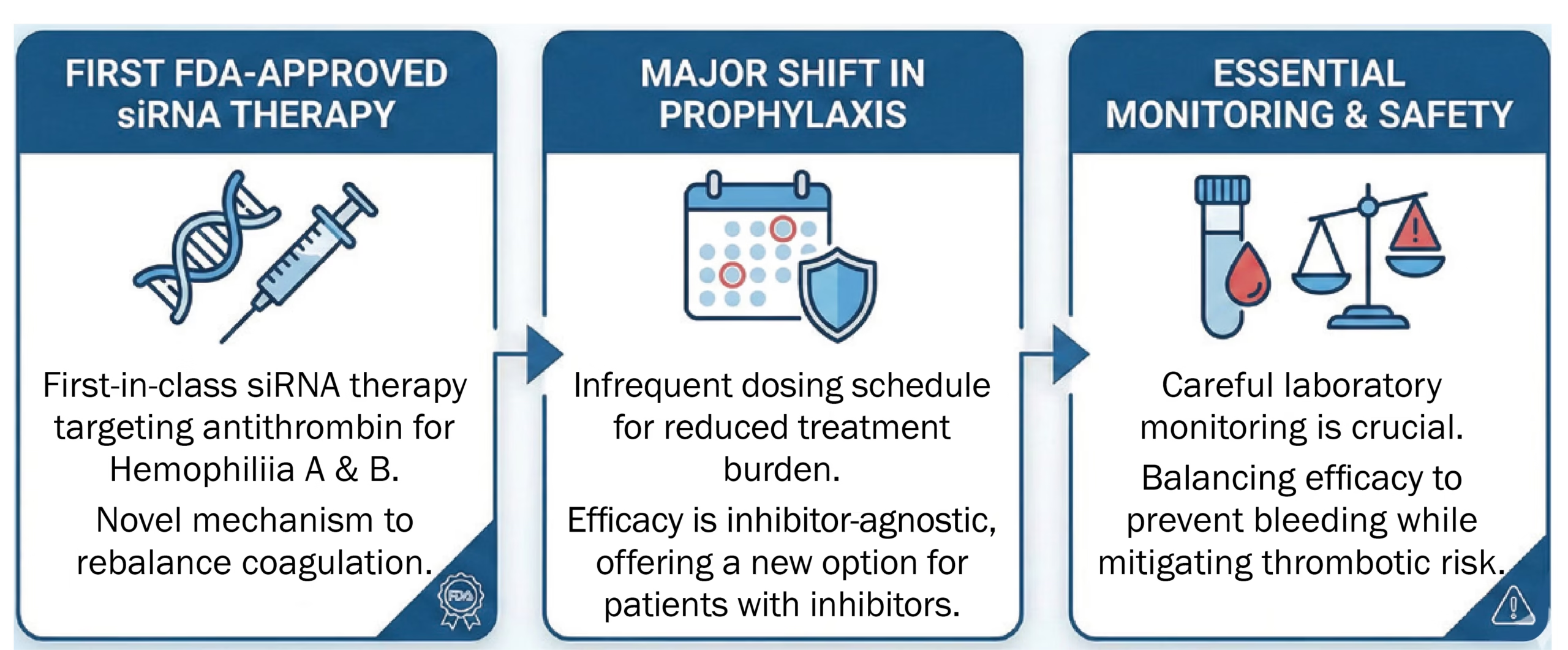
gallbladder disease, along with the need for regular liver function and antithrombin level assessments.
Source: U.S. Food and Drug Administration. FDA Approves Novel Treatment for Hemophilia A or B, with or without Factor Inhibitors (Qfitlia (fitusiran)); March 28, 2025. https://www.fda. gov/news-events/press-announcements/fda-approves-novel-treatment-hemophilia-or-b-or-without-factor-inhibitors)

At SunAct, we remain dedicated to tracking and sharing global advances that continue to redefine the landscape of cellular therapy and oncology. Stay tuned for our next edition as we uncover more breakthroughs and emerging trends shaping the future of cancer research.
Disclaimer: This newsletter is intended for healthcare professionals and researchers. Information is for educational purposes only.