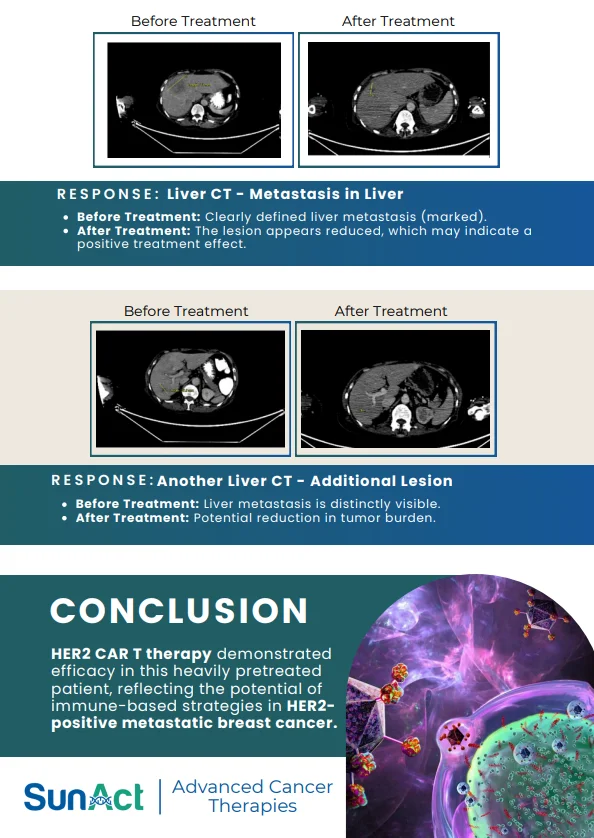
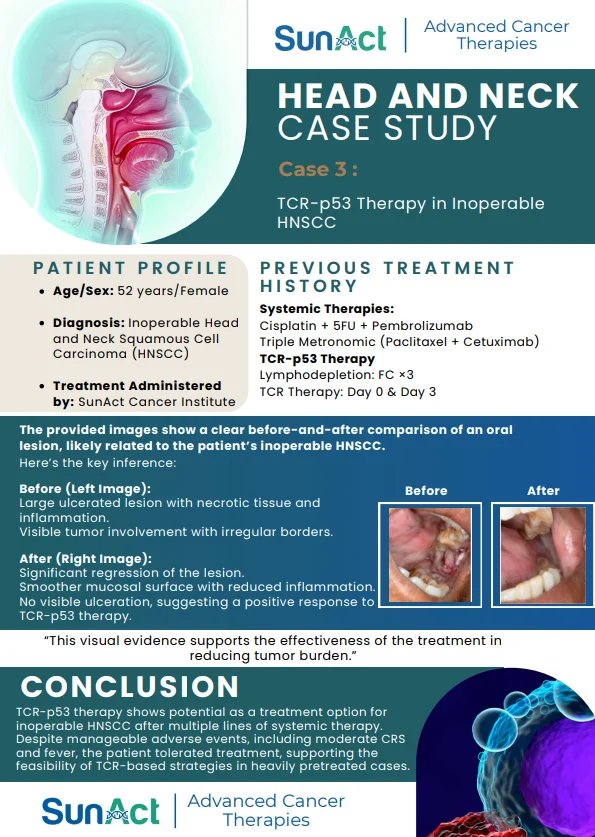
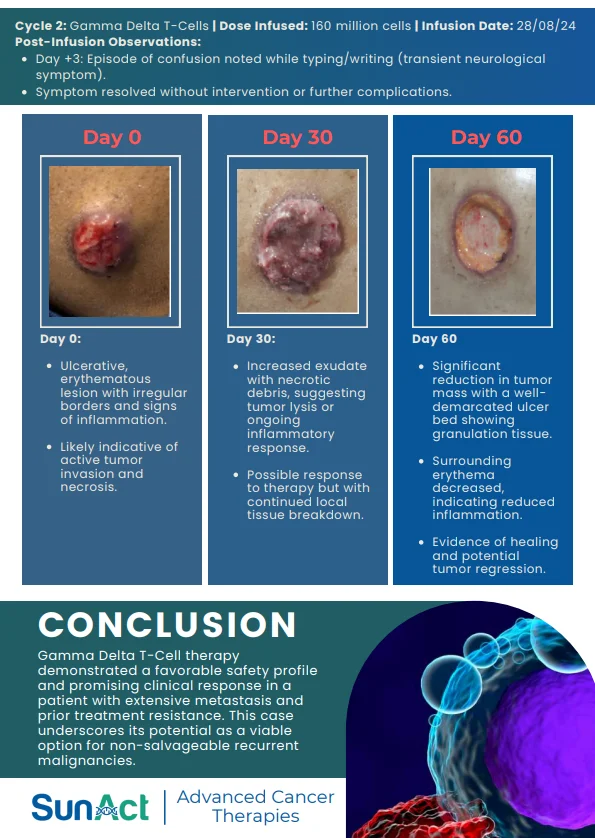
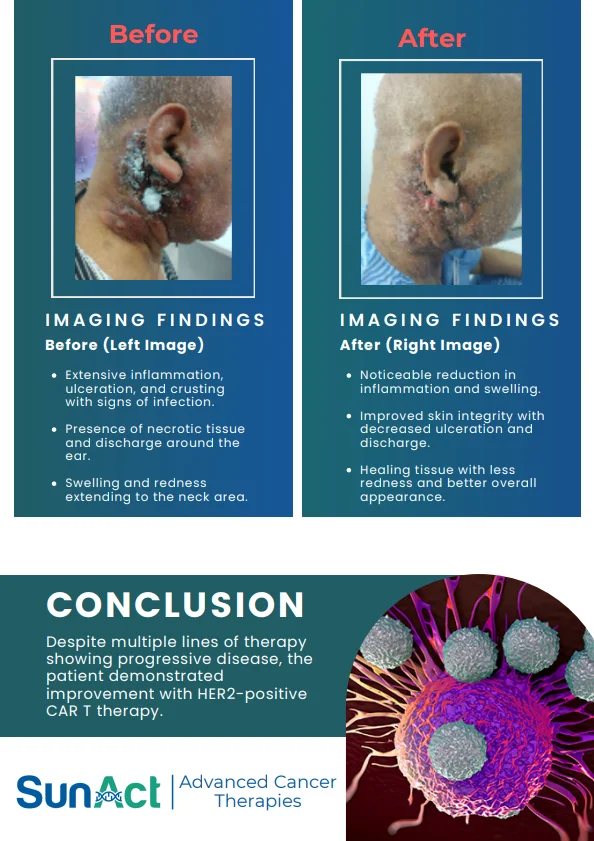
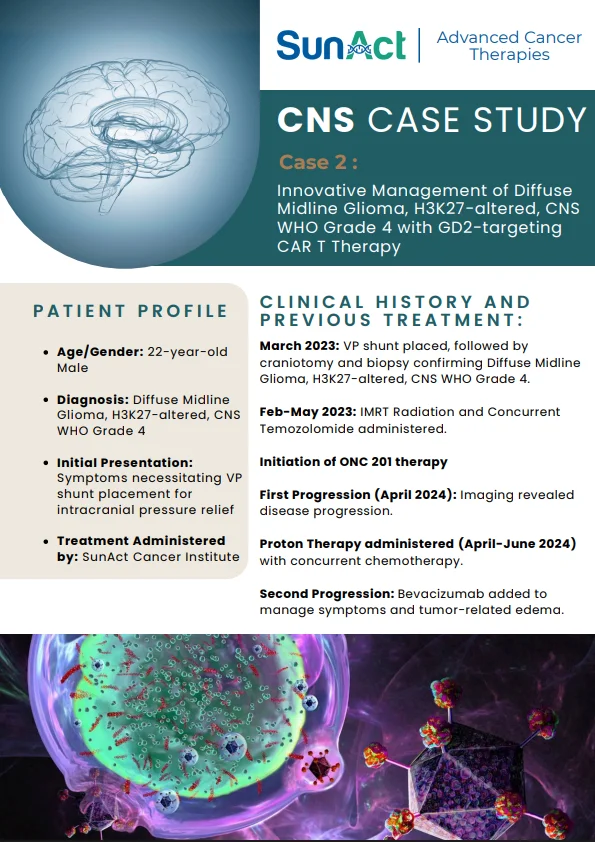
Base-Edited Anti-CD7 CAR-T | Anitocabtagene Autoleucel Trial | Single-Cell MRD in AML | FDA Approval of Lisocabtagene Maraleucel
A first-in-human Phase I study has evaluated the safety and feasibility of universal base-edited anti-CD7 CAR-T cells (BE-CAR7) in pediatric (≤16 years) and adult patients with relapsed or refractory T-cell acute lymphoblastic leukemia (T-ALL). BE-CAR7 T cells were engineered using base
editing to generate triple knockouts of TCRαβ, CD52, and CD7, enabling fratricide resistance, allogeneic use, and compatibility with alemtuzumab-based lymphodepletion. Following lymphodepletion with fludarabine, cyclophosphamide, and alemtuzumab, 11 patients received BE-CAR7. Patients achieving remission by day 28 proceeded to allogeneic hematopoietic stem-cell transplantation (allo-HSCT)
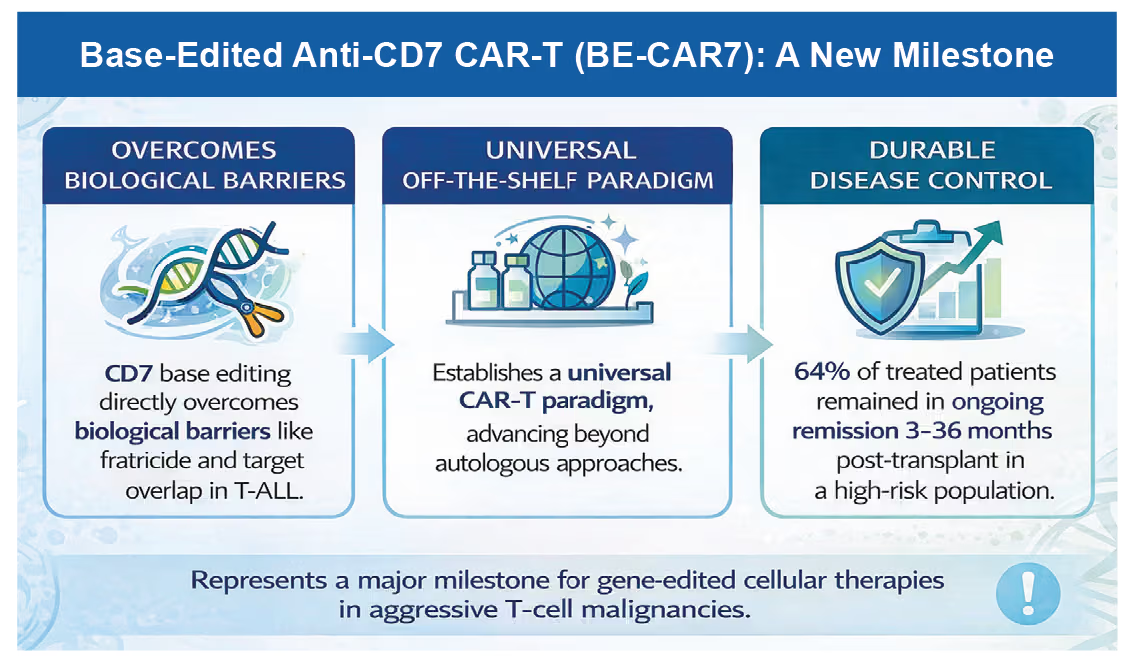
Reference: Chiesa R, Georgiadis C, Rashed H, et al. N Engl J Med. 2026;394:152–165.
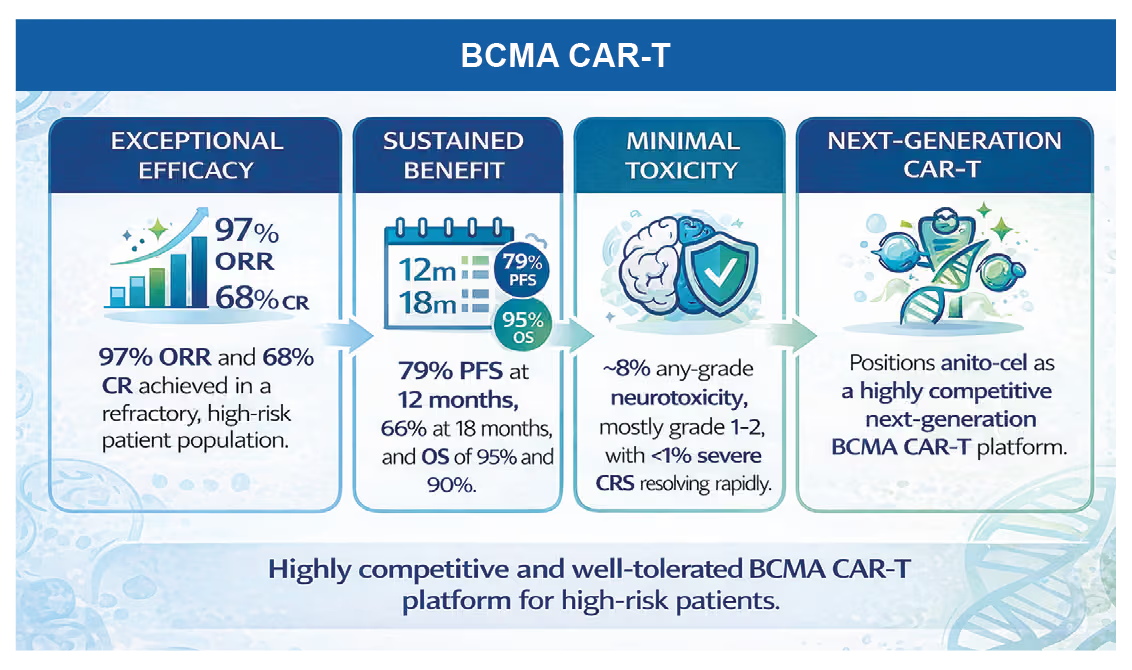
Anitocabtagene autoleucel (anito-cel) is an investigational BCMAdirected CAR-T therapy showing strong clinical promise in relapsed/
refractory multiple myeloma. Updated Phase II iMMagine-1 data
presented at ASH 2025 reported a 97% overall response rate and
68% complete response rate in a heavily pretreated population.
Anito-cel demonstrated durable progression-free and overall survival. A
key differentiator is the use of a novel synthetic BCMA-binding protein,
rather than a conventional antibody-derived scFv, which may enable
simpler manufacturing and reduced toxicity.
Reference: Patel K. Phase 2 registrational study of anitocabtagene autoleucel for the treatment of patients with relapsed and/or refractory multiple myeloma: Updated results from iMMagine—1. MD Anderson Cancer Center, Houston, TX, United States. ClinicalTrials.gov Identifier: NCT05396885
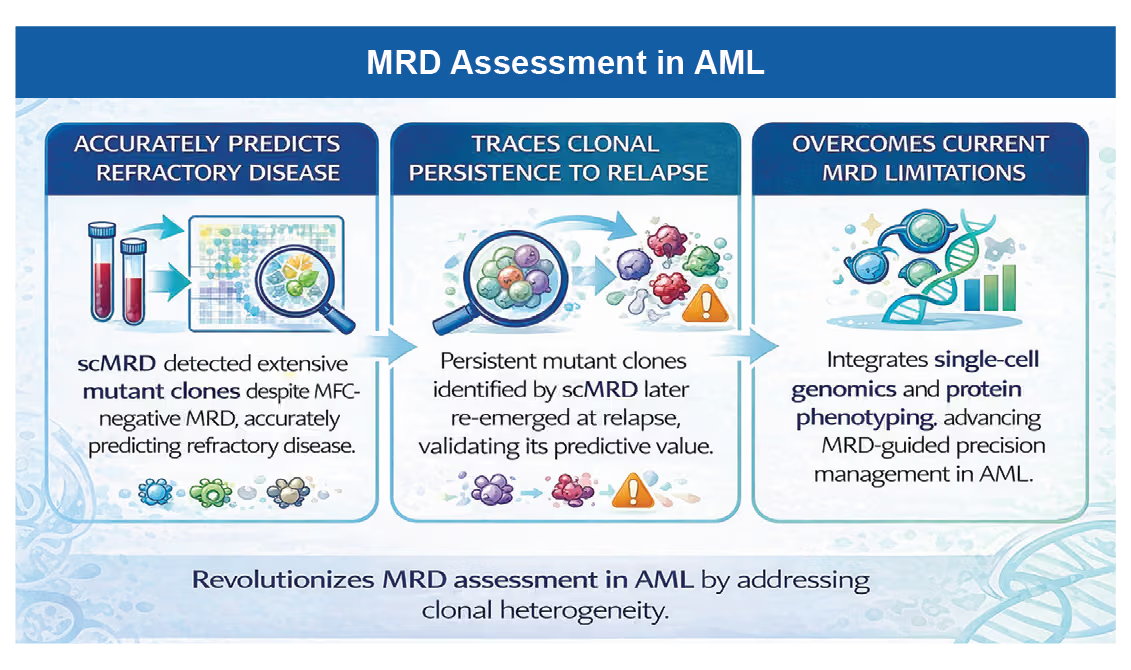
A prospective real-world study evaluated the prognostic utility of a singlecell measurable residual disease (scMRD) assay in 21 AML patients treated with standard induction chemotherapy or HMA + venetoclax. Using a single cell multiomics platform with enrichment for CD34+/ CD117+ cells, 66,515 cells were profiled and directly compared withconventional MRD methods (flow cytometry and bulk NGS).
Reference: Micin K, Madarang E, Nong T, Affer M. Blood. 2025;146(Suppl 1):280.
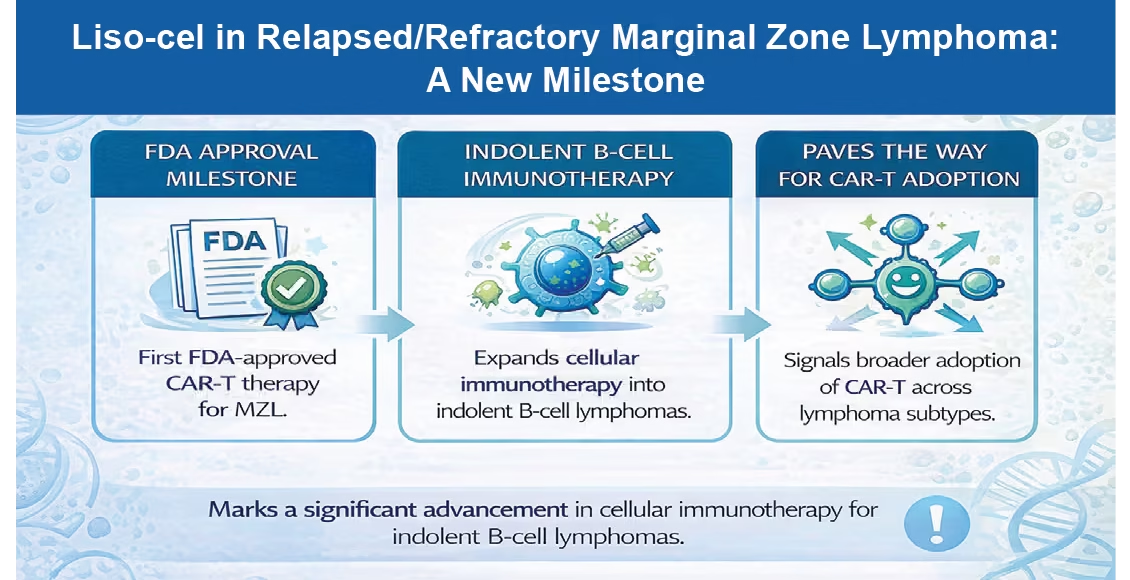
The FDA approved lisocabtagene maraleucel (liso-cel; Breyanzi) for
adults with relapsed or refractory marginal zone lymphoma (MZL) after at least two prior systemic therapies, based on the Phase II TRANSCEND FL trial.
Reference: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-lisocabtagene-maraleucel-relapsed-or-refractory-marginal-zone-lymphoma. December 4, 2025

At SunAct, we remain dedicated to tracking and sharing global advances that continue to redefine the landscape of cellular therapy and oncology. Stay tuned for our next edition as we uncover more breakthroughs and emerging trends shaping the future of cancer research.
Disclaimer: This newsletter is intended for healthcare professionals and researchers. Information is for educational purposes only.