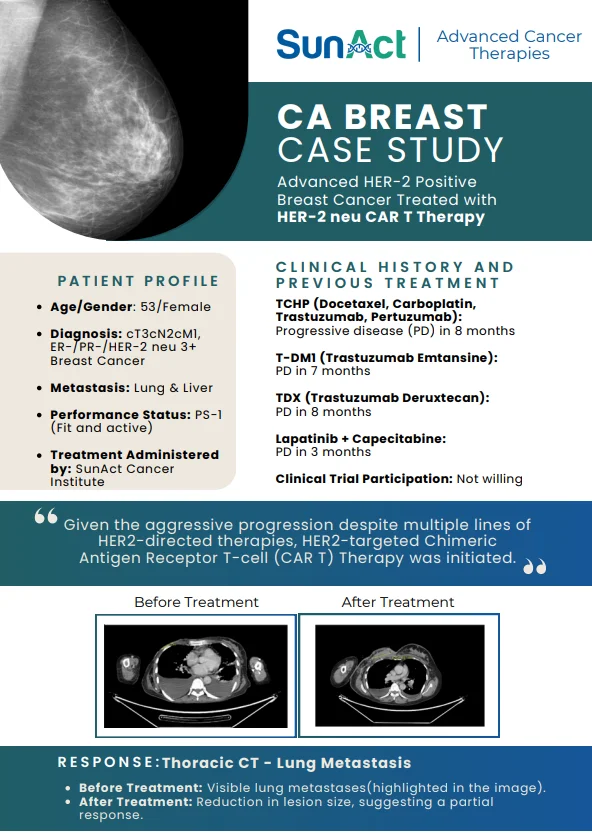
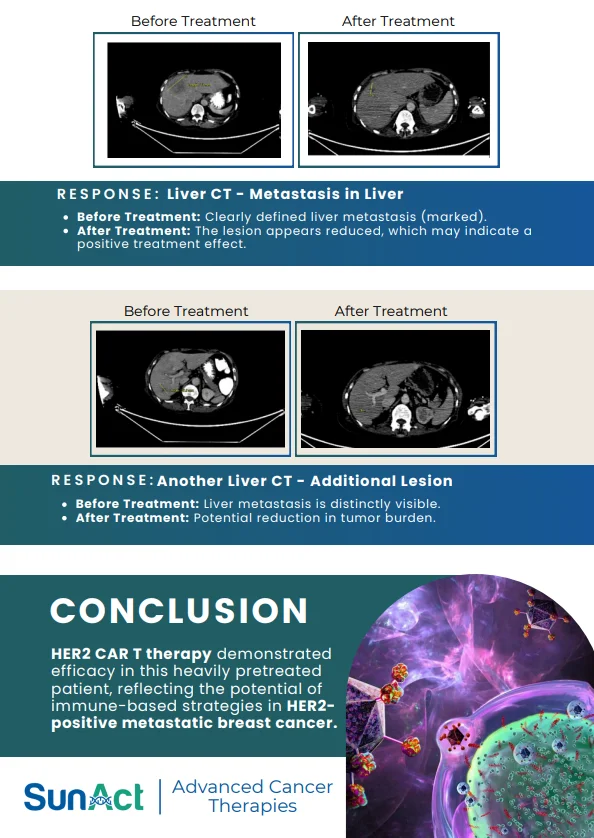
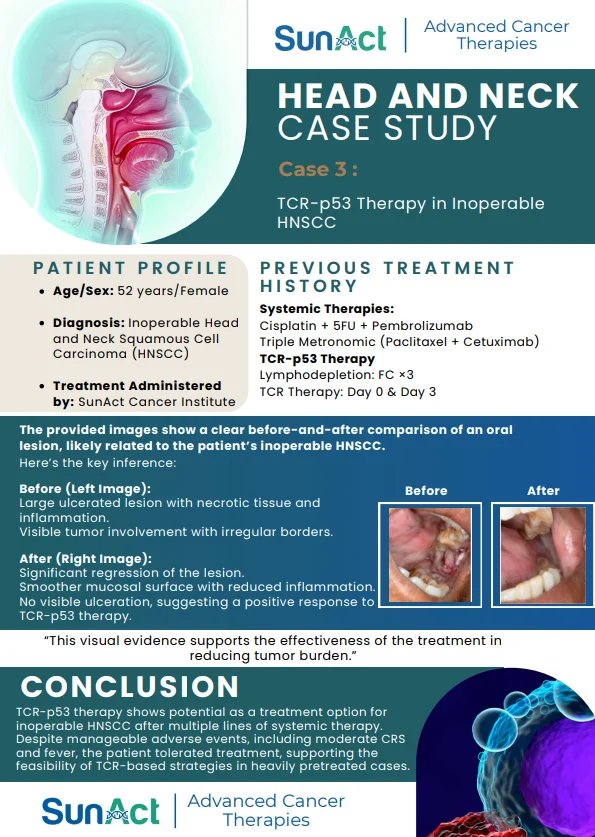
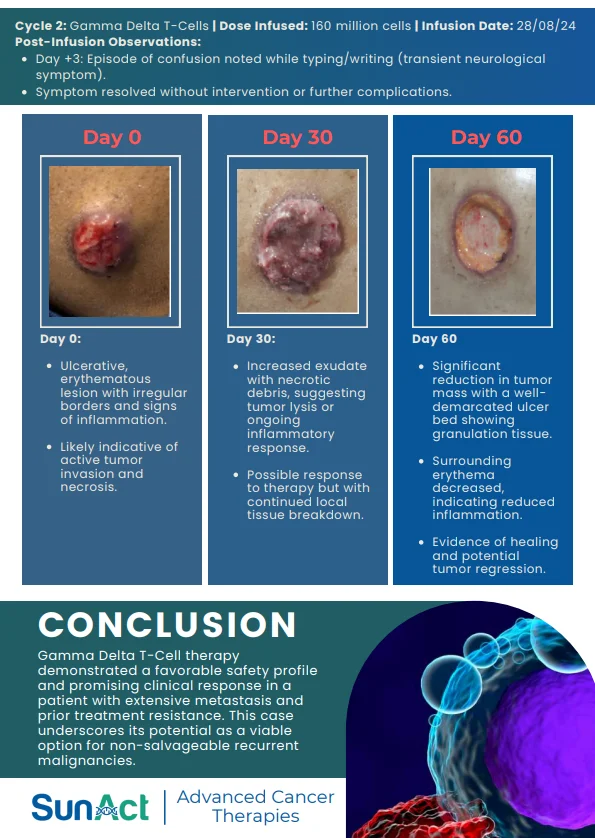
2 December 2025
A Special Treatment Option for Select Patients
Metastatic renal cell carcinoma (kidney cancer) is usually treated with targeted therapies and immune checkpoint inhibitors. While these treatments help control the disease, only a few therapies can make the cancer disappear completely. High-Dose Interleukin-2 (HD-IL-2) is one of the rare treatment options that has shown the potential for long-term, treatment-free remission in a small but important group of patients.
Over the past three decades, major international cancer centres — including the National Cancer Institute (USA) — have reported that a subset of patients receiving HD-IL-2 can experience remission lasting 10–15 years or more. Because of these durable results, HD-IL-2 remains a meaningful treatment choice even today, especially for patients who have limited curative alternatives.
HD-IL-2 is offered as a specialised immunotherapy option for carefully selected patients.
How HD-IL-2 Works?
HD-IL-2 boosts the body’s immune system by strongly activating T-cells and natural killer (NK) cells. This heightened immune response can sometimes lead to deep tumour shrinkage or complete disappearance of cancer. Since the treatment can temporarily affect blood pressure, kidney function, and fluid balance, it is administered in a controlled hospital environment with continuous monitoring.
Who May Benefit From HD-IL-2?
HD-IL-2 is suitable only for a small percentage of patients, usually those who:
- Have clear-cell renal cell carcinoma
- Have cancer limited to a few metastatic sites
- Have excellent performance status (ECOG 0–1)
- Are younger or middle-aged with good heart, lung, and kidney function
- Can undergo ICU-level monitoring during treatment
In this select subgroup, outcomes can be significant, with long-term remission achievable in some patients.
Real-World Outcomes
Long-term studies have shown:
- About 20% of patients respond to treatment
- Nearly 1 in 10 may achieve a complete response
- Most complete responses last many years
- Treatment-related deaths are extremely rare when given in experienced centres
These results make HD-IL-2 one of the few systemic therapies capable of producing durable, treatment-free remission in advanced kidney cancer.
HD-IL-2 Treatment Approach
HD-IL-2 is provided as a specialised, carefully supervised immunotherapy for eligible patients. Each case is evaluated individually to ensure maximum benefit and safety.
For selected patients, HD-IL-2 may provide a real opportunity for long-term remission.
HD-IL-2 Treatment for Kidney Cancer in India: Why SunAct HD-IL-2 therapy requires ICU-level monitoring, a dedicated oncology team, and experience managing complex immune responses
Not all hospitals in India are equipped to administer this treatment safely. At SunAct, HD-IL-2 is offered as a specialised immunotherapy option for carefully selected patients with metastatic renal cell carcinoma (RCC). Each patient is individually evaluated by our team, led by Dr. Kritika Mehrotra (Consultant, Medical Oncology), to confirm eligibility and ensure treatment is delivered with appropriate monitoring and support. Patients seeking kidney cancer treatment in India — including from Mumbai, Pune, and other cities — have consulted SunAct for access to this otherwise limited therapy
How Does HD-IL-2 Compare to Other Kidney Cancer Treatments?
Most metastatic kidney cancer patients in India are treated with targeted therapies (such as sunitinib or pazopanib) or immune checkpoint inhibitors (such as nivolumab or pembrolizumab). These treatments can control the disease and extend survival, but complete and durable remission without ongoing treatment is rare. HD-IL-2 is different: while only a small percentage of patients respond, those who do can achieve long-term, treatment-free remission lasting a decade or more. This makes it a uniquely valuable option for the right patient — one who is young, fit, has clear-cell RCC, and limited metastatic sites. For patients who do not meet HD-IL-2 eligibility criteria, SunAct’s oncology team will recommend the most appropriate targeted or immunotherapy regimen for kidney cancer treatment in India.
Frequently Asked Questions: HD-IL-2 for Kidney Cancer in India
Yes. HD-IL-2 treatment for metastatic kidney cancer (RCC) is available at SunAct in Mumbai, delivered under specialist supervision with ICU-level monitoring.
Approximately 20% of selected patients respond to HD-IL-2. Around 8–10% achieve a complete response, with many remissions lasting 10 years or more.
Eligibility requires clear-cell RCC, good performance status (ECOG 0–1), limited metastatic sites, and strong heart, lung, and kidney function. Each case is assessed individually at SunAct.
Treatment is given in cycles with close hospital monitoring. Each cycle requires a hospital stay of several days. The number of cycles depends on response and tolerance, reviewed after each round.
HD-IL-2 is a form of immunotherapy — a cytokine therapy that broadly stimulates the immune system, unlike checkpoint inhibitors which work by releasing immune checkpoints. Both are immunotherapy options for kidney cancer but suit different patients