26 March 2026
A diagnosis of stage 4 lung cancer is understandably overwhelming. Patients and families often ask the same questions at the outset: What exactly does stage 4 mean? Is it treatable? Can it be cured? What is the expected survival? And how much does treatment cost in India?
The reassuring reality is that stage 4 lung cancer treatment has changed dramatically over the last decade. It is no longer approached as one uniform disease. Instead, treatment is increasingly guided by the molecular profile of the tumour, the PD-L1 status, the patient’s general condition, and the burden of metastatic disease. For many patients, this has translated into better symptom control, longer survival, and improved quality of life.
Understanding Stage 4 Lung Cancer
Stage 4 lung cancer means that the cancer has spread beyond the lung to distant parts of the body such as the other lung, pleura, bones, liver, adrenal glands, or brain. In most patients, this is considered a systemic disease, so treatment is usually based on medicines that work throughout the body rather than surgery alone.
The two main types of lung cancer are:
- Non-small cell lung cancer (NSCLC), which is the commoner type
- Small cell lung cancer (SCLC), which is usually more aggressive
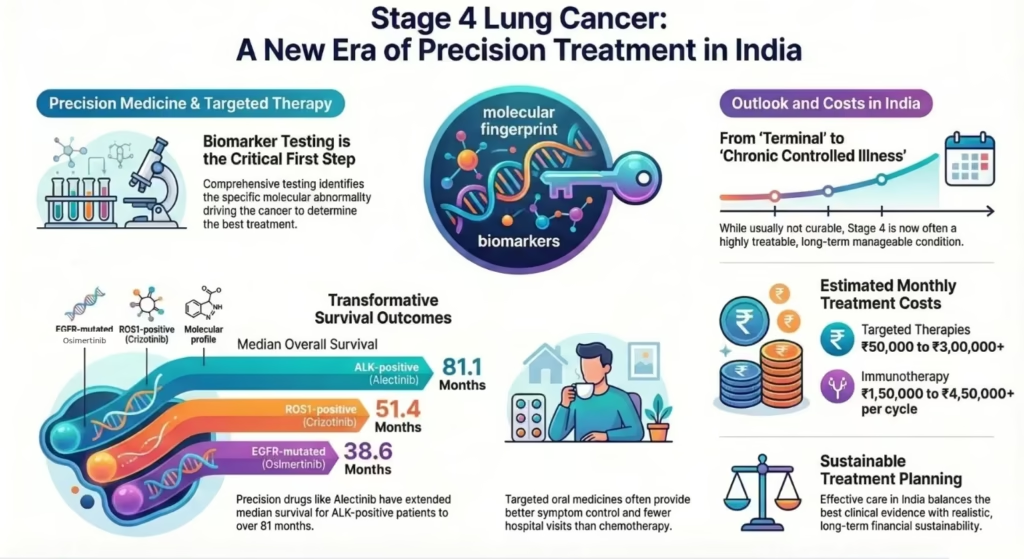
In stage 4 disease, the key goals of treatment are to control the cancer, relieve symptoms, prolong survival, and preserve quality of life. The first and most important step is to obtain the correct biopsy and perform comprehensive biomarker testing, because the treatment may differ completely depending on what molecular abnormality is driving the cancer.
Why Molecular Profiling Matters
Modern metastatic NSCLC treatment is built around testing for actionable alterations such as:
- EGFR
- ALK
- ROS1
- BRAF V600E
- MET exon 14 skipping
- RET fusion
- NTRK fusion
- KRAS G12C
- PD-L1
This is crucial because a patient with a targetable mutation often does better with a matched oral targeted therapy than with standard chemotherapy. In those without an actionable driver mutation, immunotherapy, alone or with chemotherapy, may offer durable benefit.
Treatment of Stage 4 Lung Cancer by Molecular Profile
EGFR-mutated lung cancer
In metastatic NSCLC with EGFR exon 19 deletion or L858R mutation, the preferred first-line targeted therapy is usually Osimertinib. In the phase III FLAURA trial, Osimertinib improved median progression-free survival (PFS) to 18.9 months versus 10.2 months with standard first-generation EGFR TKIs, and final analysis showed a median overall survival (OS) of 38.6 months versus 31.8 months.
For patients who develop T790M-positive resistance after earlier EGFR TKIs, the AURA3 trial established Osimertinib as standard, with a median PFS of 10.1 months and an objective response rate of 71%, clearly superior to platinum-pemetrexed chemotherapy.
Other EGFR-targeted drugs that may be encountered in practice include gefitinib, erlotinib, afatinib, and dacomitinib, though Osimertinib remains the major current standard in most suitable first-line settings.
ALK-positive lung cancer
For ALK-rearranged metastatic NSCLC, the major frontline options are alectinib, brigatinib, and lorlatinib. The final overall survival analysis of the phase III ALEX trial reported a median OS of 81.1 months with alectinib versus 54.2 months with crizotinib. That is an extraordinary figure in metastatic lung cancer and reflects how transformative precision treatment has been in this subgroup.
Lorlatinib is another highly CNS-active option, especially relevant in patients with brain metastases, and has produced remarkably durable disease control in long-term follow-up studies. In the 5-year update of the phase III CROWN study, median progression-free survival was still not reached with lorlatinib, compared with about 9.1 months with crizotinib, and the 5-year progression-free survival rate was 60% with lorlatinib versus 8% with crizotinib.
Crizotinib was the first major ALK inhibitor to significantly change outcomes in this disease. In the phase III PROFILE 1014 trial, first-line crizotinib achieved an objective response rate of 74% and a median progression-free survival of 10.9 months, clearly outperforming chemotherapy. However, one of its key limitations is relatively weaker CNS penetration, which matters because ALK-positive lung cancer has a strong tendency to involve the brain.
ROS1-positive lung cancer
For ROS1 fusion-positive NSCLC, the commonly used targeted therapies are crizotinib and entrectinib. In updated PROFILE 1001 data, crizotinib achieved a median PFS of 19.3 months and a median OS of 51.4 months.
Repotrectinib is a next-generation ROS1/TRK inhibitor designed to overcome certain resistance mutations and improve intracranial activity. In the phase I/II TRIDENT-1 study, among TKI-naive ROS1-positive NSCLC patients, repotrectinib achieved a confirmed objective response rate of 79%, with median duration of response of 34.1 months and median progression-free survival of 35.7 months. In patients previously treated with one ROS1 TKI and no chemotherapy, median progression-free survival was about 9 months.This makes repotrectinib an important modern option, particularly when CNS disease or ROS1 resistance mutations are a concern.
Entrectinib is particularly useful when CNS activity is important, and it is an important option in patients with brain metastases.
RET fusion-positive lung cancer
For RET fusion-positive NSCLC, the key targeted agents are selpercatinib and pralsetinib. In the updated LIBRETTO-001 analysis, selpercatinib showed an objective response rate of 83% in treatment-naive patients and 62% in previously treated patients, with a median duration of response of 28.6 months and median PFS of 24.9 months. In patients with measurable CNS metastases, the intracranial response rate was 85%.
MET exon 14 skipping mutation
For MET exon 14 skipping NSCLC, the major targeted therapies are capmatinib and tepotinib. In the VISION study, tepotinib demonstrated an objective response rate of 51.4%, a median duration of response of 18.0 months, a median PFS of 11.2 months, and a median OS of 19.6 months.
This is a particularly important mutation to detect because it may be missed if testing is incomplete.
BRAF V600E-mutated lung cancer
For BRAF V600E-mutated metastatic NSCLC, the standard targeted regimen is dabrafenib plus trametinib. Updated phase II data showed an objective response rate of 68.4% in pretreated patients and 63.9% in treatment-naive patients. Median PFS was 10.2 months and 10.8 months, while median OS was 18.2 months and 17.3 months, respectively.
NTRK fusion-positive lung cancer
For the rare subset of NTRK fusion-positive lung cancer, the targeted therapies are larotrectinib and entrectinib. These are tumour-agnostic drugs, meaning they work across cancer types if the fusion is present. Because NTRK fusions are uncommon in lung cancer, they are best identified through broad-panel NGS. Durable responses have been reported with both agents, making this an important though rare actionable subgroup.
KRAS G12C-mutated lung cancer
In patients with KRAS G12C mutation, important targeted drugs include sotorasib and adagrasib. These are mainly used after progression on earlier standard therapy and represent another important example of how metastatic lung cancer treatment is increasingly biomarker-driven.
Immunotherapy in Stage 4 Lung Cancer
If there is no actionable driver mutation, immunotherapy becomes central, especially in patients with significant PD-L1 expression. The most established and globally used drugs in stage 4 lung cancer include pembrolizumab, atezolizumab, nivolumab-ipilimumab & durvalumab.
Pembrolizumab: still the main foundation in metastatic NSCLC
For patients with PD-L1 expression of 50% or higher, the phase III KEYNOTE-024 trial established pembrolizumab monotherapy as a standard first-line option. At 5-year follow-up, median overall survival was 26.3 months and the 5-year overall survival rate was 31.9%. The objective response rate was 46.1%, which was clearly superior to chemotherapy.
In KEYNOTE-189 for metastatic non-squamous NSCLC, pembrolizumab plus pemetrexed-platinum achieved 5-year overall survival rates of 19.4% versus 11.3% compared with chemotherapy alone, with an objective response rate of 48.3% versus 19.9%. In KEYNOTE-407 for metastatic squamous NSCLC, pembrolizumab plus chemotherapy achieved 5-year overall survival rates of 18.4% versus 9.7%.
Real-world practice has broadly supported pembrolizumab-based treatment. In one recent real-world study of first-line pembrolizumab monotherapy for PD-L1-high metastatic NSCLC, the objective response rate was 36.4%, with median progression-free survival of 9.7 months and median overall survival of 17.0 months. This is an important reminder that while trial results are often stronger than real-world outcomes, meaningful long-term benefit is still seen outside clinical trials.
Atezolizumab: important in both NSCLC and SCLC
Atezolizumab remains an important PD-L1 inhibitor in lung cancer.
In metastatic NSCLC with high PD-L1 expression and no EGFR or ALK alteration, atezolizumab monotherapy is an option in selected patients. It is also relevant in metastatic non-squamous NSCLC in certain combination regimens, especially where a bevacizumab-containing approach is appropriate.
Atezolizumab also remains one of the best-known immunotherapy options in extensive-stage small cell lung cancer, where adding it to carboplatin-etoposide improved survival compared with chemotherapy alone.
Nivolumab plus ipilimumab, with or without short-course chemotherapy, is an option in metastatic NSCLC without EGFR or ALK alterations, particularly when a dual-checkpoint strategy is being considered.
Durvalumab is best known in lung cancer for stage III consolidation after chemoradiotherapy, but in stage 4 extensive-stage SCLC, durvalumab plus platinum-etoposide remains an important first-line option.
Second- and Third-Line Treatment in Refractory Disease
Another important part of stage 4 lung cancer care is planning for what comes after first-line treatment. When the cancer progresses, treatment decisions should ideally be guided by the molecular profile, prior therapies, pattern of progression, performance status, and repeat tissue biopsy or liquid biopsy where feasible. This matters because resistance mechanisms can sometimes reveal a new actionable target and change the next line of treatment.
In common EGFR-mutated NSCLC after progression on first-line osimertinib, platinum-pemetrexed chemotherapy remains an important backbone. The phase III MARIPOSA-2 study showed that adding amivantamab after osimertinib progression improved progression-free survival compared with chemotherapy alone.
For EGFR exon 20 insertion-mutated NSCLC, amivantamab has an even more established role. In the phase III PAPILLON trial, amivantamab plus carboplatin-pemetrexed in the upfront setting improved progression-free survival compared with chemotherapy alone. In patients already treated with platinum-based therapy, amivantamab monotherapy remains important based on CHRYSALIS, where it showed a clinically meaningful response rate and duration of response.
This distinction is useful in practice:
- In classic EGFR exon 19 deletion or L858R disease, amivantamab is most relevant after osimertinib progression, particularly with chemotherapy
- In EGFR exon 20 insertion, it is relevant both upfront with chemotherapy and after platinum as monotherapy
After repeat biopsy or NGS at progression, a small but important subgroup may be found to have a HER2 activating mutation. In this setting, trastuzumab deruxtecan (T-DXd) is an important HER2-directed option in previously treated metastatic NSCLC and remains one of the key drugs to know in HER2-mutant lung cancer. More recently, zongertinib, an oral HER2-selective tyrosine kinase inhibitor, has also emerged as an important option for HER2 activating mutation–positive non-squamous NSCLC. It is important to be precise here: these drugs are relevant for HER2-mutant disease, not for HER2 amplification alone. If repeat biopsy shows HER2 amplification rather than a clear HER2 activating mutation, management becomes more individualised and discussion of HER2-directed therapy or a clinical trial becomes particularly important.
In more heavily pretreated or third-line refractory settings, options become more individualised. Depending on prior treatment exposure, histology, organ function, and patient fitness, commonly used drugs may include docetaxel, docetaxel plus ramucirumab, gemcitabine, weekly paclitaxel, vinorelbine, or a pemetrexed-based regimen if it has not already been exhausted. In biomarker-selected refractory disease, additional targeted strategies may also become relevant after repeat NGS identifies a new alteration.
Finally, once a patient reaches a refractory stage after standard targeted therapy, chemotherapy, or immunotherapy, it is very important that the patient and family actively discuss clinical trial options with their oncology team. This is especially relevant after osimertinib progression, after platinum failure in EGFR exon 20 insertion disease, or when repeat biopsy identifies a rarer alteration such as HER2 activating mutation or another emergent resistance pathway. In modern lung cancer care, clinical trials are often the route through which patients access the next meaningful line of treatment.
Real-World Survival and Quality of Life
One of the biggest changes in stage 4 lung cancer is that survival is no longer a single fixed number. It varies greatly by molecular subtype.
A patient with EGFR-mutated disease treated with first-line osimertinib may have a median survival of 38.6 months in the FLAURA trial. An ALK-positive patient treated with alectinib in ALEX had a median OS of 81.1 months. A ROS1-positive patient treated with crizotinib in PROFILE 1001 had a median OS of 51.4 months. These are striking numbers when one considers that metastatic lung cancer historically carried a much shorter survival.
Equally important is quality of life. Many patients on well-matched targeted therapy can experience better control of cough, breathlessness, pain, appetite loss, and fatigue, often with fewer hospital visits than with conventional chemotherapy. The purpose of treatment is not only to lengthen life, but also to help the patient live better.
Cost of Stage 4 Lung Cancer Treatment Per Month in India
The cost of treatment in India varies widely depending on whether the patient receives oral targeted therapy, immunotherapy, chemotherapy, or a combination, and whether originator or generic formulations are used.
A broad practical estimate is:
- Targeted therapy: approximately ₹50,000 to ₹3,00,000+ per month
- Immunotherapy: approximately ₹1,50,000 to ₹4,50,000+ per month/cycle
- Chemotherapy: usually lower than immunotherapy, but total cost depends on regimen, day-care charges, supportive medicines, and complications
Patients also need to factor in the cost of:
- biopsy and pathology
- molecular testing / NGS
- PET-CT, CT, MRI brain
- blood tests
- supportive medications
- management of pleural effusion, infections, pain, or breathlessness
These figures vary by hospital, city, brand selection, and frequency of treatment. In India, affordability is an important part of treatment planning, and a good oncology discussion includes not only the best evidence-based option but also realistic, sustainable choices.
Many patients and families are not aware that patient assistance and patient support programs may be available for selected targeted therapies and immunotherapy drugs. In addition, many centres also have access to hospital-based financial counselling teams, charitable support mechanisms, social work departments, and government-linked health schemes for eligible patients.
Can Stage 4 Lung Cancer Be Cured in India?
This is the most emotionally important question for most patients and families.
In most cases, stage 4 lung cancer is not considered curable in the classical sense. However, it is very much treatable, and in selected patients it can be controlled for several years. With targeted therapy and immunotherapy, some patients now live long enough for the disease to behave more like a chronic controlled illness rather than an immediately terminal one.
So the honest answer is: usually not curable, but often highly treatable, and in many patients increasingly controllable with meaningful quality of life.
Final Thoughts
Stage 4 lung cancer treatment in India has entered a far more precise and hopeful era. The best treatment is no longer chosen by stage alone, but by the molecular fingerprint of the cancer. This is why biomarker testing before treatment is essential.
For some patients, osimertinib, alectinib, lorlatinib, selpercatinib, tepotinib, dabrafenib-trametinib, larotrectinib, or other targeted therapies may offer the best chance of prolonged disease control. For others, pembrolizumab or other immunotherapy-based combinations may be the right choice. The goal is to combine evidence-based treatment with practical decision-making, affordability, supportive care, and dignity.
In refractory settings, especially after progression on osimertinib or after exhaustion of standard therapy, repeat biopsy, repeat molecular testing, and discussion of clinical trial options can be just as important as the next prescription itself.